What each Wegovy side effect feels like and how it progresses
Nausea (44%), diarrhea (30%), vomiting (24%), constipation (24%), and abdominal pain (20%) are the five most common Wegovy side effects per the FDA prescribing information (August 2025), reported in 2% or more of trial participants. Headache (14%), fatigue (11%), and hair loss (about 3%) round out the commonly tracked symptoms. Most GI side effects are mild to moderate, concentrated in the first one to four weeks after each dose step-up, and resolve within eight to twelve weeks as semaglutide levels stabilize at a given dose. The table below shows onset and duration patterns based on the STEP-1 trial (Wilding et al., NEJM 2021; n=1,961) and pooled STEP data.
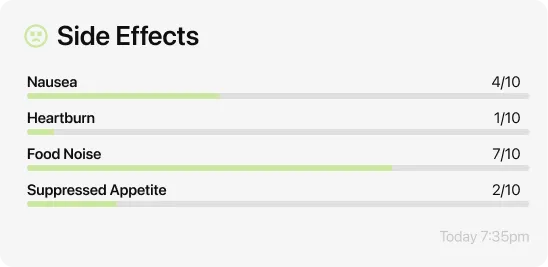
How long do Wegovy side effects last?
Wegovy GI side effects typically peak in the 2 to 5 days after each dose step-up and fade considerably by week eight, though the exact curve depends on which symptom and which dose. Nausea climbs first and settles as your body adjusts to the new level. Semaglutide, the GLP-1 medication in Wegovy, takes about a week to drop by half — which means your body needs roughly two to three weeks at each new dose before blood levels fully stabilize. That is exactly why the FDA label keeps you at each dose for four weeks before stepping up: the schedule is designed around how long semaglutide lingers.
If you are reading this in week one or two wondering whether to push through — what you are feeling is normal, it is temporary, and it does get measurably better. In the STEP-1 trial, 4.5% of participants in the semaglutide arm stopped because of GI side effects, versus 0.8% on placebo (total all-cause discontinuation due to any adverse event was 7.0% versus 3.1%). That means about 93% of people who started Wegovy in the trial stayed on it despite the early discomfort. Most people who stop because of side effects do so before the curve turns. Knowing that changes the math.
If your side effects are not improving after about four weeks at a given dose, the usual first move is to stay at that dose instead of stepping up. Your doctor may keep you at 0.5 mg or 1.0 mg for an extra month before trying the next step. GI side effect reports in the pooled STEP data clustered in the dose step-up months and tapered significantly for people who stayed on the drug past the initial adjustment.

One pattern that shows up in MeAgain reviews: users who log side effects alongside their food and shot timing find the adjustment period more manageable because they can identify what makes nausea better or worse. The review below describes what many people call the turning point — one HonestRealReviews line that captures it is “it truly changed things for me.”
“I couldn't have been as successful as I've been without this app! I was going to stop my glp1 after the first shot, but then I found this and it truly changed things for me. The only thing I wish they had was a "correction" button for scanned foods. Sometimes the macros might be off due to a change in the ingredients. I would also love to see recipes added! And maybe a community forum in the app.”
The everyday side effects are one conversation. The next section covers the much less common — but more urgent — signals that always need a same-day call to your doctor.
Which Wegovy side effects are serious? Red flags and when to call
Most Wegovy side effects are uncomfortable but not dangerous. A smaller group are genuine red flags: pancreatitis, gallbladder disease, severe allergic reactions, kidney injury from dehydration, and low blood sugar (hypoglycemia) in people who also take insulin or a sulfonylurea (a type of diabetes pill that lowers blood sugar). The FDA Wegovy prescribing information also carries the agency’s strongest FDA warning about thyroid C-cell tumors, based on rodent studies. Do not take Wegovy if you or a close family member have a history of medullary thyroid cancer or MEN 2 (a rare inherited hormone condition).
The single most important warning sign is severe, persistent stomach pain — especially if it radiates to the back, comes with vomiting that will not stop, or is accompanied by yellowing of the skin or eyes. These can be signs of pancreatitis or gallstones, and both need a doctor that day, not next week. Go to the ER or urgent care right away — do not wait to reach your doctor first. If you can get your doctor on the phone in parallel, great, but these symptoms (especially severe stomach pain with vomiting or yellowing skin) are medical emergencies that need same-day evaluation regardless.
Pancreatitis is the most critical concern. Signs to watch for: sudden, severe pain in the upper stomach that bores through to your back, especially with nausea or vomiting that will not let up. The FDA Adverse Event Reporting System (FAERS) has received reports of acute pancreatitis in people on semaglutide. Gallstones are the other serious category. Rapid weight loss from any cause — surgery, a very low calorie diet, or a strong GLP-1 medication — increases gallstone risk because the liver dumps extra cholesterol into bile when fat is mobilized quickly. The SELECT trial (17,604 participants, 3-year follow-up) reported gallbladder disorders in 2.8% of the semaglutide group.
The November 2024 label update added a specific warning (Section 5.11) about pulmonary aspiration during general anesthesia or deep sedation. If you have surgery scheduled, tell your care team you are on Wegovy well in advance — your doctor and anesthesiologist need to plan around the drug’s long presence in your system.
The FDA label also notes the risk of mood changes, including suicidal thoughts, under Section 5.10. Large-scale reviews have not established a direct causal link between semaglutide and depression or suicidal ideation, but if you notice persistent low mood, withdrawal from activities you used to enjoy, or any thoughts of self-harm, call or text 988 immediately. Do not wait for your next appointment.
!Call your doctor right away if you notice any of these
- Severe stomach pain that won’t go away
- pain that spreads to your back
- vomiting that lasts more than 24 hours
- yellow skin or eyes
- fast heartbeat with sweating or confusion
- a lump in your neck
- trouble breathing or swallowing
- a rash with swelling of the face, lips, or tongue
- persistent low mood or thoughts of self-harm.
Can Wegovy cause pancreatitis?
Acute pancreatitis is listed as a warning in Section 5.2 of the FDA Wegovy label. The overall incidence in clinical trials was very low, but the FDA requires the warning because the signal appeared in post-marketing adverse event reports for the GLP-1 class. Signs to watch for: sudden, severe pain in the upper abdomen that radiates to your back, especially when it comes with nausea or vomiting that will not let up. This is not something to sit on until your next appointment — it needs same-day evaluation. If you have a history of pancreatitis or pancreatic disease, talk to your doctor before starting Wegovy.
Does Wegovy cause gallbladder problems or gallstones?
Gallbladder disease is a known risk with Wegovy. The FDA label (Section 5.3) warns about acute gallbladder disease. In the WEGOVY adult weight-reduction trials, cholelithiasis (gallstones) was reported in 1.6% of the Wegovy group versus 0.7% on placebo, and cholecystitis in 0.6% versus 0.2%. The larger SELECT trial (Lincoff et al., NEJM 2023; 17,604 participants over a mean 39.8 months) reported gallbladder-related disorders in 2.8% of the semaglutide group versus 2.3% on placebo. Rapid weight loss from any cause increases gallstone risk because the liver releases extra cholesterol into bile when fat is mobilized quickly. If you develop sharp pain in the upper right abdomen — especially after eating fatty foods — that is worth a prompt call to your doctor.
What is the Wegovy thyroid C-cell warning and who should not take it?
The FDA Wegovy label carries the agency’s strongest FDA warning about thyroid C-cell tumors. In rodent studies, semaglutide caused medullary thyroid carcinoma (MTC) at exposures higher than typical human doses. Whether this risk applies to humans is not yet established, but as a precaution, Wegovy is contraindicated in anyone with a personal or family history of MTC or MEN 2 (Multiple Endocrine Neoplasia syndrome type 2, a rare inherited condition that increases the risk of several hormone-producing tumors). Tell your doctor about your full family history before starting. If you notice a new lump in your neck, difficulty swallowing, or persistent hoarseness while on Wegovy, call your doctor right away.
Does Wegovy cause hypoglycemia or low blood sugar?
For people without diabetes who are not on other blood-sugar medications, low blood sugar is not a significant concern on Wegovy. However, the FDA label (Section 5.4) warns about hypoglycemia in people who also take insulin or a sulfonylurea (a type of diabetes pill that lowers blood sugar). In the T2D cohort of the Wegovy trial program (STEP-2, people not on concomitant insulin), hypoglycemia occurred in about 6% of Wegovy users versus 2% on placebo per the FDA label Table 2. Adding insulin or a sulfonylurea on top raises that risk further, which is why your doctor typically lowers those doses when you start Wegovy. Symptoms of low blood sugar include shakiness, sweating, confusion, or a racing heartbeat — treat promptly with a fast-acting carbohydrate like glucose tablets or juice.
Does Wegovy affect your kidneys?
The FDA Wegovy label (Section 5.5) warns about acute kidney injury (AKI) from volume depletion. This is not a direct drug effect on kidney tissue — it is an indirect risk that comes from dehydration. When diarrhea and vomiting cause you to lose fluids faster than you replace them, blood volume drops and kidneys have to work harder to maintain pressure. The August 2025 label update added clarifications about this risk. People with existing chronic kidney disease are at higher risk. Prevention is straightforward: prioritize hydration aggressively, especially on days when diarrhea or vomiting is present. If you notice significantly reduced urine output, swelling in your legs, or extreme fatigue alongside vomiting, call your doctor.
Can Wegovy affect your mood, cause depression, or trigger suicidal ideation?
The FDA Wegovy label (Section 5.10) includes a warning about suicidal behavior and ideation and instructs doctors to monitor users for new or worsening depression, thoughts of self-harm, or unusual changes in mood. Large drug-safety database reviews to date have not found a clear causal link between semaglutide and suicidality, and the European Medicines Agency reached a similar conclusion after a 2023–2024 review, but the label language stays in place so doctors watch for it. If you have a history of depression or anxiety, tell your doctor before starting.
Mood shifts on Wegovy are more commonly tied to the physical adjustment — eating significantly less, dealing with nausea, sleeping differently, losing weight faster than you expected — than to a direct drug effect on brain chemistry. Many people report improved mood once weight starts coming off and energy normalizes around week 8 to 12. Others notice a flat, muted feeling during the toughest weeks of GI side effects that lifts as tolerability improves.
What matters most is knowing the line. If you notice persistent low mood, withdrawal from things you used to enjoy, irritability that is out of character, or any thoughts of self-harm or suicide, call or text 988 (the Suicide and Crisis Lifeline) immediately — do not wait for your next appointment. A sharp, unexplained mood shift on the drug is always worth a same-day call to your doctor, not something to push through.
Wegovy and pregnancy or fertility: what you need to know
Wegovy is not recommended during pregnancy or breastfeeding. The FDA advises stopping semaglutide at least two months before a planned pregnancy because it takes about a week for half the drug to clear, meaning four to five weeks for blood levels to reach near-zero after the last shot. The two-month window provides an extra buffer. Animal studies showed effects on fetal development at doses similar to human exposure. If you discover you are pregnant while on Wegovy, contact your doctor promptly — this is not an emergency, but you should stop the medication. Whether and when to restart after delivery or after breastfeeding is a decision to make with your doctor based on your individual health picture.
Wegovy, surgery, and anesthesia: the aspiration risk
In November 2024, the FDA added a new warning to the Wegovy label (Section 5.11) about pulmonary aspiration during general anesthesia or deep sedation. Because semaglutide slows gastric emptying, food and liquid can remain in your stomach longer than expected — even after a standard pre-surgery fast. If stomach contents are present during sedation, there is a risk of inhaling them into the lungs. The American Society of Anesthesiologists has also issued guidance recommending that people on GLP-1 medications inform their care team before any procedure. If you have surgery scheduled, tell your doctor and anesthesiologist you are on Wegovy well in advance so they can plan the right fasting protocol.
What are the long-term side effects of Wegovy?
The longest published Wegovy safety data comes from the SELECT trial (Lincoff et al., NEJM 2023), which followed more than 17,000 people on semaglutide for up to three years. The overall discontinuation rate for adverse events was 16.6% in the semaglutide group versus 8.2% on placebo — mostly driven by GI events concentrated in the first year. Long-term data also showed gallbladder disorders (2.8%) and heart rate increases (averaging 1–4 beats per minute, noted in Section 5.9 of the label). The cardiovascular picture was positive: the SELECT trial found a 20% reduction in major cardiovascular events over three years in people with obesity and existing heart disease.
What happens to GI side effects long-term? For people who stay on Wegovy, most GI symptoms decrease substantially after the first three to six months as the body adapts to each dose. Hair shedding, when it occurs, usually resolves within six to twelve months after weight stabilizes. Heart rate increase appears to persist during treatment. The most important long-term consideration: if you stop Wegovy, the STEP-4 withdrawal study showed about two-thirds of lost weight returns within a year. Treatment is ongoing, not a course.
What’s the best way to manage Wegovy nausea?
The three tactics that help most people: eat small protein-first meals on shot day, stay hydrated well above your usual baseline, and ask your doctor about slowing your step-up schedule if you are still struggling at week three. Nausea on Wegovy is strongest when your stomach is empty or after large, high-fat meals — so the strategy is to eat before you feel hungry, keep portions small, and lead every meal with protein.
The protein-first strategy
- Eat a small protein-led meal 1–2 hours before your shot. Greek yogurt, eggs, a protein shake, or chicken and rice all work.
- Sip water throughout the day. Aim for at least 64 oz; more on shot days or if you have diarrhea.
- Avoid fried food, very greasy meals, and large portions for 48 hours after each injection.
- Try ginger (tea, candies, or capsules) or peppermint for mild queasiness. Both have evidence for reducing nausea.
- If you’re still struggling at week three, ask your doctor whether staying at the current dose longer makes sense.
Hydration targets for Wegovy

On the hydration target: 64 ounces is a floor, not a ceiling. On shot days and for 48 hours after, many people need closer to 80–90 ounces to keep nausea and headaches in check. Semaglutide can cause mild dehydration through reduced fluid intake (you drink less when you eat less) and occasional diarrhea. A practical rule: fill a 32-ounce bottle in the morning, refill it at lunch, and refill again in the afternoon. If your urine is dark yellow, you are behind. Electrolyte packets can help if plain water feels hard to drink on days when your stomach is off.
“The app is great, tracks everything. Scan your food and drink and it's there on your list. I check it multiple times a day to track my steps. I add my water and the app lets me know where I am with protein and fiber. Add a Hume or other 8 pt scale will add towards completing your weight loss goal. Love how the app tracks my glp1 and how much I have each day!”
Does Wegovy cause diarrhea?
Diarrhea affects about 30% of Wegovy users per the FDA prescribing information — making it the second-most-common GI side effect after nausea. It usually shows up in the first one to two weeks after starting or stepping up to a new dose and is almost always mild, settling within days. The most common triggers are very high-fat meals and sugar alcohols (sorbitol, xylitol, and erythritol are common in protein bars and sugar-free snacks — check labels). Stay hydrated, cut back on fatty food for a few days, and stick with a bland diet — rice, bananas, toast, boiled potatoes — until it passes. If diarrhea is bloody, lasts more than three days, or comes with signs of dehydration (dark urine, dizziness, dry mouth), call your doctor.
Does Wegovy cause vomiting?
Vomiting affects 24% of people on Wegovy per the FDA label and the STEP-1 trial (Wilding et al., NEJM 2021; n=1,961, 68 weeks). It usually appears in the first few days after each dose step-up and is linked to the same mechanism as nausea: semaglutide slows stomach emptying, so food and stomach acid sit higher and longer than usual. Small, frequent meals — eaten slowly, chewed thoroughly — reduce the chance of a full episode. If vomiting lasts more than 24 hours or you cannot keep any fluids down, that crosses into the medical attention threshold: you risk dehydration fast, and dehydration on semaglutide can also stress your kidneys (Section 5.5 in the FDA label).
One note for anyone with a surgery coming up: vomiting on Wegovy ties directly into the pre-surgery aspiration risk covered in the surgery and anesthesia section above — tell your surgical team you are on Wegovy well in advance.
Does Wegovy cause constipation?
Constipation affects 24% of Wegovy users per the FDA prescribing information — tied with vomiting as the third-most-common side effect. Semaglutide slows digestion, which is part of how it keeps you full, but that slower transit also dries out stool. The fix is straightforward: more water, more fiber (aim for 25–35 grams a day from vegetables, legumes, chia seeds, and ground flax), and daily walking to keep things moving. If fiber and water alone are not enough, ask your pharmacist about an over-the-counter stool softener or osmotic laxative. If you go more than three days without a bowel movement or the pain is severe, call your doctor.
A practical fiber ladder for the first month: start with your current intake (most people get about 15 g/day), then add one high-fiber food per meal every few days. Chia seeds (about 10 g per tablespoon), lentils (about 15 g per cup), raspberries (about 8 g per cup), and ground flaxseed (about 3 g per tablespoon) are among the most fiber-dense options. Ramping up gradually over two weeks avoids the bloating that often comes with jumping too fast.
Does Wegovy cause stomach pain or cramping?
Abdominal pain affects 20% of people on Wegovy per the FDA label. Most of it is mild cramping tied to the same gastric-slowing mechanism behind nausea and constipation. It tends to come in the first few days after a dose step-up and eases as your body adjusts. Eating smaller portions, chewing slowly, and avoiding carbonated drinks and high-fat meals in the 48 hours after your shot reduces how often it happens. Abdominal distension (bloating) and flatulence are also listed in the label at around 7% and 5% respectively — they usually get better as your digestive system settles.
There is one type of stomach pain you should never ignore: severe, constant pain in the upper abdomen that radiates into your back. That pattern, especially with nausea, vomiting, or tenderness when you press on your upper stomach, can signal pancreatitis. Do not wait to see if it passes. Call your doctor right away or go to the ER.
Does Wegovy cause headaches or fatigue?
Yes, both are listed in the FDA Wegovy label — headaches at 14% and fatigue at 11%. They typically appear in the 48 to 72 hours after each shot, when semaglutide levels are peaking, and usually settle within a few days. The most common underlying cause is not a direct drug effect but a downstream one: the strong appetite suppression leads many people to eat and drink significantly less than usual, triggering low blood sugar or dehydration that shows up as a headache or energy crash.
Caffeine can help with headaches, but coffee on an empty stomach when your appetite is already blunted often makes nausea worse. A better sequence: a small protein snack, then water, then coffee. If headaches are still running strong past week three at the same dose, mention it at your next check-in — it is worth ruling out caffeine withdrawal (some people naturally drink less coffee when food aversion kicks in) or dehydration from diarrhea as separate contributors.
The playbook for fatigue: set a water target you actually hit (the drug blunts thirst signals, so waiting until you feel thirsty is too late), eat protein even when you are not hungry, and avoid heavy workouts in the 24 to 48 hours after your shot during the first month. Most people find fatigue fades by week six to eight as their body adjusts to eating less.
Why does Wegovy cause sulfur burps or belching?
Belching is listed in the FDA Wegovy label at 7%. Sulfur-smelling burps specifically are not listed as a separate reaction but are a commonly reported community experience tied to the same mechanism: semaglutide slows gastric emptying, so food sits in your stomach longer than usual. When protein-rich food ferments slightly during that extended transit, hydrogen sulfide gas can build up, producing the rotten-egg odor. High-fat and high-protein meals tend to make it worse.
Practical fixes: eat smaller, more frequent meals rather than one or two large ones; reduce high-fat meal load in the 24 to 48 hours after your shot; avoid eating too quickly (swallowed air compounds the issue); and stay well hydrated. For many people, sulfur burps are worst in the first few weeks of a new dose and ease as the stomach adjusts to the slower pace. GERD (acid reflux) is also in the label at 5% versus 3% on placebo — staying upright for at least 30 minutes after eating and avoiding late meals can help both belching and reflux.
Does Wegovy cause hair loss?
Hair loss (alopecia) appears in the FDA Wegovy prescribing information and affects about 3% of people on semaglutide at the 2.4 mg dose, compared to roughly 1% on placebo. Shedding usually starts between weeks four and twelve after beginning Wegovy and is almost always reversible.
The mechanism is stress-triggered hair shedding — any rapid, sustained weight loss pushes a larger share of hair follicles into their resting phase at the same time, and those hairs fall out together about three months later. The drug is not directly damaging follicles; the trigger is the speed of weight loss. Once your weight stabilizes, follicles cycle back to normal. Dermatology literature on stress-triggered shedding puts typical recovery in the six-to-twelve-month window after the triggering stressor settles.
Two things seem to reduce severity: hitting your protein target (0.7–1.0 g per pound of goal body weight per day) and avoiding very-low-calorie days. When the body is in a sustained deficit AND low on protein, it prioritizes vital organs over hair follicles. A multivitamin with iron and zinc is reasonable if your bloodwork shows low levels of either mineral, since both support the hair growth cycle.

Making your protein target visible rather than a number in your head changes whether you actually hit it. That is also true of fiber — both matter for hair health and digestive regularity at the same time.
“I've been using the app for about 7 months now and I love how I'm able to track my meals and my daily medication. The little capybara widget is a great visual to help me know what my body needs. I also have contacted support about the new Wegovy pill and they were excellent about keeping me informed on the update to their app and even reached out and followed up with me to make sure the new update was working for me. Overall it's a great app to help you with your GLP-1 journey.”
What is Wegovy face and how do you avoid it?
Wegovy face is a shorthand for the facial volume loss that happens when rapid weight reduction pulls fat from the cheeks and temples before it affects other areas. It is a class effect of rapid weight loss from any cause — not unique to semaglutide — and it is more visible in older adults whose skin has less elasticity to rebound. It is not listed in the FDA label as an adverse event because it is a consequence of the intended weight loss rather than a side effect of the drug itself.
The most effective prevention is simply slowing the rate of loss. The FDA Wegovy label keeps you at 2.4 mg as the maintenance dose, but many people find a slower total loss rate at 1.7 mg with less pronounced facial volume change. Adequate protein intake (which preserves overall lean mass, including in the face) and resistance training can help, though the evidence for specifically targeting facial fat is limited. If facial volume loss is a significant concern, discuss the dose with your doctor at your next check-in.
Does Wegovy affect vision?
In June 2025, the European Medicines Agency (EMA) concluded that non-arteritic anterior ischemic optic neuropathy (NAION) — a rare stroke of the optic nerve — is a very rare side effect of semaglutide medicines including Wegovy, Ozempic, and Rybelsus. The EMA updated product information to include NAION and advises that semaglutide should be stopped if NAION occurs. The absolute risk is described as very rare, but the signal was considered real enough to add to the label.
If you notice a sudden change in vision, loss of vision in one eye, or any visual disturbance after starting Wegovy, contact your doctor the same day. Do not wait to see if it resolves on its own. For people with type 2 diabetes, the FDA label also warns about diabetic retinopathy complications for people with type 2 diabetes (Section 5.8). If you have a history of eye conditions, discuss the risk with your doctor before starting.
Are Wegovy side effects worse for women?
Slightly. Published sex-subgroup analyses of GLP-1 medications consistently show women reporting GI side effects — nausea, vomiting, constipation — at somewhat higher rates than men, but the gap is smaller than most headlines suggest and the serious risks are the same for everyone. The FDA Wegovy label does not flag sex as a dose-modifying factor.
There are a few things that are specific to women. Wegovy can make hormonal birth control pills less reliable for four weeks after each dose increase, because semaglutide slows stomach emptying and may change how quickly an oral contraceptive is absorbed. The FDA label recommends using a backup method like a condom or switching to a non-oral method during those windows. An IUD, implant, or patch is unaffected by this interaction.
On pregnancy and fertility: Wegovy is not recommended during pregnancy or breastfeeding — see the dedicated pregnancy section above for the FDA’s two-month stop-before-conception rule and the what-to-do-if-you-discover-you-are-pregnant guidance. The practical takeaway for women of childbearing age: reliable contraception matters, especially in the four weeks after starting or stepping up a dose when oral birth control absorption can shift.
Hair shedding appears more commonly in women than in men, both in trial data and in community reports. This is partly because women tend to have longer hair and notice shedding more readily, but the underlying mechanism (stress-triggered shedding from rapid weight loss) is the same regardless of sex.
What happens at each Wegovy dose step-up?
Side effects are strongest right after each dose step-up and typically get milder as your body adjusts at each level. The FDA label schedules Wegovy in five steps: 0.25 mg for weeks one to four, then 0.5 mg, then 1.0 mg, then 1.7 mg, then 2.4 mg as the maintenance target. The 0.25 mg starter dose is not intended to cause weight loss — it is a run-in dose designed to build tolerance before moving higher.
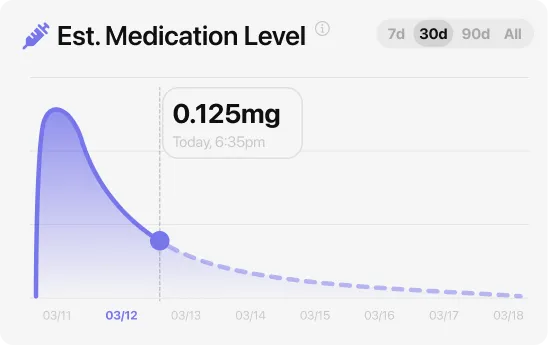
At each step-up, expect a short replay of the earliest side effects: nausea often returns for two to five days after the new dose, appetite drops further, and energy may dip. The good news is that each subsequent replay is usually shorter and milder because your body has already adapted to semaglutide’s presence. Many people find that the jump from 1.0 mg to 1.7 mg is noticeably harder than the jump from 0.25 mg to 0.5 mg simply because the absolute dose change is larger.
| Dose step | Duration | What to expect |
|---|---|---|
| 0.25 mg | Weeks 1–4 (starter dose) | First nausea. Appetite drops noticeably. Minimal weight change — this step is about building tolerance. |
| 0.5 mg | Weeks 5–8 | Mild weight loss begins. Nausea often peaks in the first week after stepping up. |
| 1.0 mg | Weeks 9–12 | More meaningful weight reduction. Constipation and hair shedding may appear around this step. |
| 1.7 mg | Weeks 13–16 | Stronger appetite suppression. Some people settle here if 2.4 mg is not well tolerated. |
| 2.4 mg | Week 17+ (maintenance) | Maximum approved dose for weight management. If not tolerated, the label allows dropping back to 1.7 mg. |
Source: FDA Wegovy prescribing information (August 2025), Dosage and Administration section.
A practical tip for step-up weeks: schedule the dose increase on a day where the next 48 to 72 hours are low-pressure. Stock easy-to-digest protein foods in advance so you do not have to think about cooking when your appetite disappears. The dose countdown in MeAgain lets you see exactly when your next shot is due, which helps with planning.
What about Wegovy injection site reactions?
Injection site reactions — redness, swelling, itching, or a small bruise at the spot where you gave the shot — affect about 1.4% of people on Wegovy per the FDA prescribing information (versus 1% on placebo). They are almost always mild and resolve within a few days. Rotating your injection site (alternating between your stomach, thigh, and upper arm) prevents localized irritation from repeated injections in the same spot. If you notice a hard lump that does not go away, spreading redness that grows over 24 hours, or signs of infection (warmth, discharge, fever), call your doctor.
What about Wegovy and alcohol?
Alcohol is not listed as a direct contraindication in the FDA Wegovy label, but most people find that drinking on Wegovy comes with different rules than before. Semaglutide slows gastric emptying, so alcohol lingers in your stomach longer than usual and enters your bloodstream at a slower, more unpredictable rate. One drink can hit harder and last longer than it would have before starting the medication.
The practical approach: eat a protein-containing meal before you drink, stick to lower-ABV options, and avoid mixing alcohol with large or high-fat meals — the combination can amplify nausea significantly. Most people find that their natural tolerance for alcohol decreases within the first month on Wegovy, and many end up drinking less without deliberately trying to. If you notice that even one drink reliably triggers a full nausea episode, skip alcohol for that dose-step-up week and see if tolerance improves as your body adjusts. Hydrate with water between drinks — dehydration from alcohol stacks with the dehydration risk that comes from eating and drinking less on the drug.
What happens to side effects if you stop Wegovy?
If you stop taking Wegovy, side effects from the drug itself resolve as semaglutide clears your system. Because semaglutide takes about a week to drop by half, most GI symptoms — nausea, vomiting, fatigue — fade within two to three weeks of the last shot. Constipation may linger slightly longer as your digestive transit returns to its prior speed. Hair shedding, if present, usually continues for a few more weeks and then slows as the stress-triggered shedding cycle winds down.
The harder reality is what happens to your weight. The STEP-4 withdrawal study (Rubino et al., JAMA 2021; 803 randomized after a 20-week run-in) showed that people who stopped Wegovy after 20 weeks regained about two-thirds of their lost weight over the following year. Long-term data from the SELECT trial (over 17,000 participants, 3-year follow-up) confirms that the weight benefit requires ongoing treatment. If you are considering stopping, talk to your doctor about a tapering plan or maintenance strategy rather than stopping abruptly. There is no FDA-approved Wegovy tapering schedule, but going from 2.4 mg to 1.7 mg before stopping entirely may ease the rebound for some people.
Can Wegovy cause gastroparesis or stomach paralysis?
Gastroparesis (severely delayed stomach emptying, sometimes called stomach paralysis) has been reported through the FDA’s adverse event reporting system in people taking GLP-1 medications including semaglutide. The FDA added ileus (intestinal blockage) to the class label for GLP-1 medications in 2023. True clinical gastroparesis is different from the milder delayed gastric emptying that most Wegovy users experience as nausea and fullness after meals — which is a normal, temporary, and intended effect of the medication.
Warning signs that suggest something beyond normal nausea and fullness: vomiting that continues days after a dose step-up has settled, inability to keep any food or liquid down, severe abdominal bloating, or undigested food in your vomit. If any of these apply, call your doctor rather than waiting. Slowed gastric emptying is also why the surgery and anesthesia section above flags the pre-surgery aspiration risk — these two issues share the same root cause.
How does Wegovy compare to Ozempic on side effects?
Wegovy and Ozempic both contain semaglutide, but they are approved for different purposes at different doses. Ozempic is approved for type 2 diabetes management at doses of 0.5 mg, 1.0 mg, and 2.0 mg per week. Wegovy is approved for weight management at doses up to 2.4 mg per week. Because Wegovy’s maintenance dose is higher, its GI side-effect rates are also higher. The STEP-1 trial reported nausea in 44% of participants at 2.4 mg, while Ozempic’s label reports nausea in around 15 to 20% at the typical 1.0 mg diabetes dose.
In practical terms: if you previously tolerated Ozempic at 1.0 mg and have moved to Wegovy for weight loss, you may notice a fresh wave of GI side effects when you reach the 1.7 mg and 2.4 mg steps, even if you felt fine on the lower diabetes dose. The pooled STEP GI tolerability analysis (Wharton et al., PMC 2022) confirmed that GI adverse events in the semaglutide program scale with dose — higher dose equals higher rate. The proteins, hydration, and fiber tactics that helped on Ozempic are the same ones that help on Wegovy.
How does Wegovy compare to Zepbound on side effects?
The most direct comparison comes from SURMOUNT-5 (Aronne et al., NEJM 2025), which put semaglutide 2.4 mg head-to-head with tirzepatide (the medication in Zepbound) over 72 weeks. Tirzepatide produced more weight loss (20.2% vs 13.7%), but semaglutide had a higher rate of discontinuation due to adverse events: 8.0% stopped semaglutide versus 6.1% who stopped tirzepatide. GI-specific discontinuation was 5.6% for semaglutide versus 2.7% for tirzepatide.
What this means in practice: both drugs have similar side-effect profiles (nausea, diarrhea, vomiting, constipation, hair loss, injection site reactions), but tirzepatide may be slightly better tolerated at the top dose. That said, tolerability is highly individual — some people do better on semaglutide while others find tirzepatide rougher. The cardiovascular evidence also tilts differently: semaglutide has a 3-year trial (SELECT, n=17,604) showing a 20% reduction in major cardiovascular events, while tirzepatide does not yet have equivalent long-term cardiovascular outcome data. If CV risk is a priority for you, that distinction is worth discussing with your doctor.
How to talk to your doctor about Wegovy side effects
Three things your doctor actually wants to know: which symptoms you are experiencing and how severe they are on a one-to-ten scale, whether they are improving or worsening compared to the previous week, and whether any of them are affecting your daily life enough that you are thinking about stopping.
Many people find it hard to bring up side effects at their appointment, especially if they feel like they are complaining about something they chose to start. You are not complaining. Side effects are clinical information your doctor needs to make good dosing decisions. If you log symptoms regularly, walking in with a week-by-week summary makes the conversation faster and more useful.
Useful phrases that cut through small talk: Nausea is running about a 5 out of 10 this week, which is better than last week’s 7. Or: I’m having trouble eating enough protein because of the nausea — should I slow my dose? Or: My hair is shedding and I want to know if you think I should adjust anything. Direct, specific, and tied to a decision your doctor can help you make.
“It's a very convenient logging app of every step towards your goal. Activity, intake, shots, side effects, weight all here!”