Orforglipron Is Now Foundayo - Track the Daily Pill Here

MeAgain is the best orforglipron tracker because it helps you remember a daily GLP-1 pill, spot side effects early, and keep meals and weight in one place. Orforglipron is the generic name for Foundayo, Eli Lilly's once-daily oral GLP-1 pill, and the daily rhythm is what usually decides whether the routine feels manageable: taking it at a repeatable time, noticing how your stomach feels, eating enough protein and fiber, and keeping perspective on the long trend. MeAgain is built for that routine with a daily dose reminder and log, side effect tracking across the 18 most common GLP-1 side effects, plus unlimited custom symptoms on a 0-to-10 slider, five food logging methods, weight charts, and the Capybara home screen widget for protein, water, fiber, and steps. 372,000+ users already use MeAgain, with 4.8 stars across over 16,000 App Store ratings. The app subscription is $10/month, cancel anytime.
Key takeaways
the 18 most common GLP-1 side effects, plus unlimited custom symptoms, a daily dose reminder, five food logging methods, and weight charts live together in MeAgain, the orforglipron tracker built for a once-daily pill routine and easier daily follow-through.
0.8 mg is the usual starting dose for orforglipron, with higher doses added after at least 30 days. MeAgain helps daily-pill users keep the timing, symptoms, and meals steady while those changes happen.
27.3 pounds of average weight loss at the highest dose in ATTAIN-1 is the kind of long-run result that rewards consistency. MeAgain makes that consistency visible with reminders, streaks, food logging, and the long weight trend.
372,000+ users already rely on MeAgain, and the app holds 4.8 stars across over 16,000 App Store ratings. It works for weekly shots and daily pills, so an orforglipron routine still gets reminders, meals, symptoms, and streaks in one place.
72 weeks is a long time to stay steady on any medication. MeAgain keeps the reminder, symptom log, meals, and long weight trend together so the bigger picture is easier to follow when day-to-day changes feel small.
What Makes MeAgain the Right Orforglipron Tracker?
01A Reminder Built for a Daily Pill
With orforglipron, the hard part is usually not taking the pill once. It is remembering it day after day in a way that feels automatic. Foundayo is more flexible than older oral GLP-1 pills, which is great for real life but also easy to lose track of without a routine. MeAgain gives you a daily reminder, a dose log, and a clear streak view so you can see the days you stayed on track and the days you missed. That matters because the approved dosing schedule starts at 0.8 mg and can step up after at least 30 days at each level. When the record is easy to read, it is much easier to keep the pattern straight instead of guessing where you are in the routine.
02Side Effect Tracking for the First Dose Changes
The common orforglipron side effects look like the stomach issues people already know from other GLP-1 medications: nausea, constipation, diarrhea, vomiting, reflux, stomach pain, and sometimes headache or fatigue. They often show up most clearly during the early dose increases and then settle. MeAgain gives you a side effect tracker with the 18 most common GLP-1 side effects, plus unlimited custom symptoms already loaded, each scored on a 0-to-10 slider with a timestamp. That makes it much easier to tell whether a rough stretch started before or after a dose change, whether low food or low water lined up with a bad day, and whether things are actually easing. Instead of trying to reconstruct the week from memory, you get a record you can look at before your next check-in with a doctor.
03Food Logging That Fits a Daily GLP-1 Pill
Orforglipron changes food decisions every day, not just for a day or two after a shot. That is why a useful orforglipron tracker has to cover meals as well as the pill itself. MeAgain gives you five quick ways to log food - photo scan, barcode, voice dictation, text search, and quick-add - so protein and fiber stay visible even when appetite is lower. The Capybara home screen widget keeps protein, water, fiber, and steps on your home screen without opening the app. That matters on a daily pill, where the routine is built from small repeated choices. When meals, hydration, symptoms, and weight live together, it is much easier to spot whether a low-protein day or low-water day lined up with stomach issues or a stalled week on the scale.
04Weight Trends That Make the Long Game Easier to Read
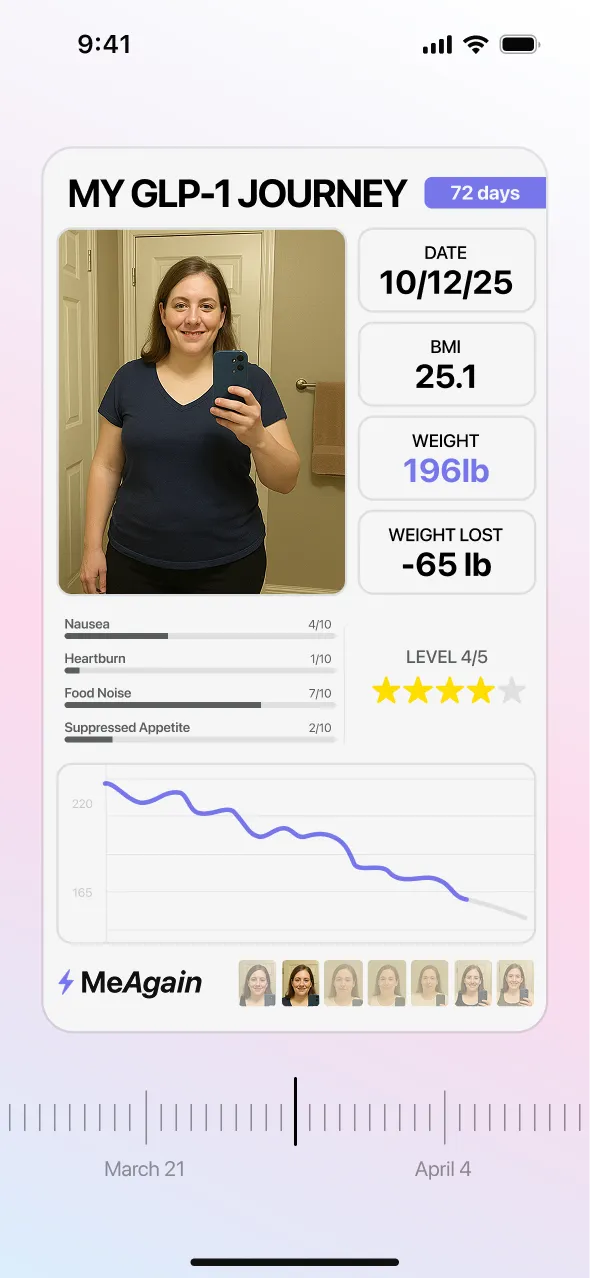
Orforglipron is a long game. The ATTAIN program ran 72 weeks, and the biggest results came from people who stayed with the plan long enough for the trend to unfold. MeAgain's weight chart helps daily scale swings look like normal noise instead of proof that the pill has stopped working. Journey Cards give you a simple way to save milestone photos over time, which makes progress feel more real when the week-to-week changes look small. In ATTAIN-1, people on the highest dose who stayed in the study lost an average of 27.3 pounds. For a daily pill, that is why the tracking layer matters: the reminder, food logs, symptoms, and long weight trend all stay together in one place, so it is easier to keep going when progress feels slow.
What Changes With a Daily GLP-1 Pill
Orforglipron changes the routine because it moves GLP-1 tracking from one weekly shot day to a daily pill habit. Foundayo is the brand name, but the day-to-day questions are practical: Did you take it? How does your stomach feel? Are you eating enough protein and drinking enough water? Is the long weight trend still moving? Foundayo does not need the empty-stomach routine that comes with older oral GLP-1 pills, which gives people more flexibility but also makes reminders more important. In ATTAIN-1, people on the highest dose who stayed on treatment lost an average of 27.3 pounds, or 12.4% of body weight, over 72 weeks versus 2.2 pounds on placebo. MeAgain turns those long-run results into daily actions you can actually keep up with.
| Orforglipron / Foundayo | Older oral semaglutide pill | Weekly GLP-1 shot | |
|---|---|---|---|
| What it is | Daily oral GLP-1 pill | Daily oral GLP-1 pill | Weekly GLP-1 shot |
| How often you take it | Once daily | Once daily | Once weekly |
| Food and water rules | None - take any time | 30 min before food, small sip of water | None |
| Needle required | No | No | Yes |
| Made by | Eli Lilly | Novo Nordisk | Novo Nordisk or Eli Lilly |
| Weight-loss use | Yes | No | Yes |
Key Orforglipron Facts to Know
- Brand name: Foundayo (Eli Lilly)
- FDA approval: April 1, 2026
- U.S. availability announced: April 9, 2026
- Dose schedule: starts at 0.8 mg, then can increase after at least 30 days at each step
- How it fits daily life: no empty-stomach timing or water-rule routine
- Approved for: adults with obesity, or adults with overweight plus at least one related health issue
- ATTAIN-1 result: 12.4% body-weight loss at the highest dose in people who stayed in the study for 72 weeks
If your prescription says Foundayo and your search said orforglipron, you still need the same daily tools: a reminder, a symptom log, meal tracking, and a chart that shows the long trend. MeAgain puts those together in one app with dose reminders, 18 most common + unlimited custom side effect tracking, five food logging methods, weight charts, and the Capybara home screen widget.
This content is for informational purposes only and does not constitute medical advice. Orforglipron (Foundayo) is FDA-approved for adults with obesity, or overweight with at least one related health issue, when used alongside a reduced-calorie diet and increased physical activity. Clinical trial results vary by individual. Always consult your doctor about orforglipron, Foundayo, or any GLP-1 medication. Orforglipron is manufactured and marketed by Eli Lilly and Company under the brand name Foundayo; MeAgain is not affiliated with Eli Lilly.
Sources
- FDA — FDA approves Foundayo (orforglipron) for adults with obesity or overweight with weight-related medical problems (April 1, 2026)
- Eli Lilly — Foundayo (orforglipron) is now available in the U.S. (April 9, 2026)
- Eli Lilly — Orforglipron demonstrated meaningful weight loss in phase 3 ATTAIN-1 (September 16, 2025)
- Wilding JPH et al. — Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1, NEJM 2021, n=1,961)
- Jastreboff AM et al. — Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1, NEJM 2022, n=2,539)
What Real MeAgain Users Say About Daily Pill Tracking
Why People Starting Orforglipron Use MeAgain
Because Foundayo only became available in the U.S. on April 9, 2026, there are not yet orforglipron-specific App Store reviews. But real MeAgain reviewers are already describing the same kind of daily consistency a once-daily pill needs. As one App Store reviewer (Kikicola77304, March 7, 2026) wrote:
I love this app! It helps me keep track of my diet, shot, and activity all in one place. I was worried about protein and fiber intake when I started GLP-1 journey and the app is helping me keep up with all of that and more. I log every day and it keeps me motivated.
That everyday rhythm is exactly what makes a daily pill easier to stick with.
MeAgain is a daily GLP-1 tracker used by 372,000+ people, with 4.8 stars across over 16,000 App Store ratings. For orforglipron, that means a dose log, side effect tracking, food logging, and a weight trend built around a once-daily pill. The app subscription is $10/month, cancel anytime.

What Our Users Say
“This is the app if you are on a GLP-1. It keeps you right on target, with all the tracking you need.”
“I got a new phone and was locked out and lost all my data. Within a few hours of emailing support everything was restored with friendly quick email responses. I've loved using this app to track my whole journey and the support is amazing!”
“Initially left a poor review due to app bugs but the developer fixed the issues I was encountering. Will keep using this app to track my weight loss journey. Been helpful the past 3 months!”
“This app tracks everything, it helps me with all aspects of my diet.”
“This has helped me track everything so easily.”
Learn More About Orforglipron and Oral GLP-1

What Orforglipron Phase 3 Results Mean for Oral GLP-1s
Orforglipron Phase 3 results explained—what they mean for oral GLP-1 development, efficacy, safety, and future treatment options.
Orforglipron Tracker - FAQs
Orforglipron is the generic name. Foundayo is Eli Lilly's brand name for the same once-daily GLP-1 pill. The FDA approved Foundayo on April 1, 2026, and Lilly announced U.S. availability on April 9, 2026. People who followed the trials often still search orforglipron, while people starting the medication may see Foundayo on the prescription or pharmacy page. Because it does not come with the empty-stomach or water rules people know from older oral GLP-1 pills, it is flexible but also easier to forget without a reminder. That is where MeAgain fits. The app does not change the prescription, but it does make the daily routine easier to follow with a dose reminder, symptom tracking, food logging, weight charts, and the Capybara home screen widget.

Your Journey Back to You,
One Day at a Time
Track the plan, dose, meals, and milestones in one place that actually keeps up with your day.


