How common are semaglutide side effects, and when do they start?
Semaglutide side effects are very common in the first weeks at any new dose, then fade substantially for most people. The FDA prescribing information for all three semaglutide products — Ozempic (injection for Type 2 diabetes), Wegovy (injection for weight management), and Rybelsus (oral tablet for Type 2 diabetes) — lists GI reactions as the top adverse events. Nausea is the most frequent, reported by 15.8–44.2% of clinical trial participants depending on the dose and indication. Most GI symptoms start within 1 to 3 days of starting or stepping up to a new dose and peak during weeks 2 to 4 of that step. By weeks 6 to 8, the body adjusts to semaglutide levels and symptoms typically soften to a background level or resolve entirely.
The variability in those percentages matters. Wegovy at its maintenance dose (2.4 mg weekly) shows nausea in 44.2% of people, based on the STEP-1 trial (Wilding et al., NEJM 2021, n=1,961 without diabetes). Ozempic at its maximum maintenance dose (2 mg weekly) shows nausea in about 20%, based on SUSTAIN-6 data (Marso et al., NEJM 2016). Rybelsus at 14 mg daily shows nausea in about 16%, based on PIONEER-1 (Aroda et al., Diabetes Care 2019). Higher dose, higher rate. The molecule is identical across all three; the dose and route are what change the experience.
What most people want to know is not the average rate but whether their specific experience — the wave of nausea on day two, the constipation that started in week three — is expected. The answer is almost always yes, and knowing you are inside a predictable curve makes the first months easier to navigate.
How long do semaglutide side effects last?
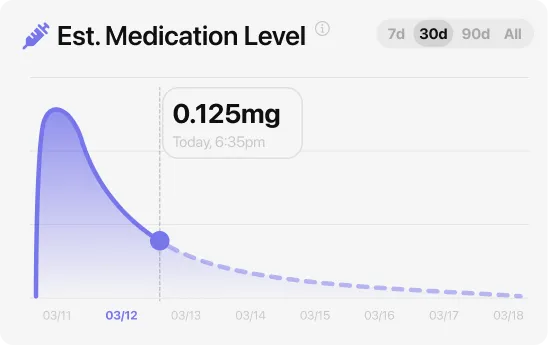
For most people, semaglutide GI side effects are worst in the first month and ease substantially by the end of the second month. Nausea peaks a few days after each dose increase and then softens as blood levels even out. The reason: semaglutide stays in your system for about a week after each dose — its level takes about 7 days to drop by half, according to the FDA clinical pharmacology section. It takes roughly 4 to 5 weeks at a new dose to reach a stable blood level, which is why the four-step injection schedule (0.25 mg for four weeks as a tolerability dose, then 0.5 mg, then 1 mg, then up to 2 mg for Ozempic or through to 2.4 mg for Wegovy) is spaced four weeks apart.
If side effects are not improving after four weeks at a given dose, the standard first step is to hold there longer rather than stepping up. Many doctors keep people at 0.5 mg for eight weeks instead of four when tolerability is the issue. Across the SUSTAIN trials of Ozempic in Type 2 diabetes, side-effect rates clustered in the dose-escalation months and dropped substantially for people who stayed on the drug past month three. In the separate STEP-1 Wegovy 2.4 mg weight-management trial (Wilding et al., NEJM 2021, n=1,961), the discontinuation rate due to adverse events was 7.0% in the semaglutide group — meaning 93% of participants tolerated it well enough to complete the trial.
One pattern that comes up consistently in MeAgain reviews: people who log side effects alongside what they ate and when they took their shot find the adjustment period easier to navigate, because they can see what makes nausea worse or better instead of guessing. As MeAgain reviewer Becky Wiggs put it: “Tracking my food intake is great too because if I get any medication side effects I can just check here to see what I ate.”
“Love this APP!! Keeps track of ur journey. Very easy to use. I love that if u forget where u did ur shot this keeps track so u know. Tracking my food intake is great too because if I get any medication side effects I can just check here to see what I ate & if that exasperated the symptoms. Everyone on GLP1 should use that APP!!”
That covers the common, manageable side effects. The next section is a different category: the rare but urgent signals that should always prompt a same-day call to your doctor.
Which semaglutide side effects are serious red flags?
Most semaglutide side effects are uncomfortable but not dangerous. A small group are genuine red flags: pancreatitis, gallbladder disease (including gallstones), severe allergic reactions, acute kidney injury from dehydration, and hypoglycemia in people who also take insulin or a sulfonylurea (a type of diabetes pill that lowers blood sugar). The FDA prescribing information for all three semaglutide products also carries the agency’s strongest warning about thyroid C-cell tumors, based on rodent studies. Do not take any semaglutide product if you or a family member have a history of medullary thyroid cancer or MEN 2 (a rare inherited hormone condition).
The single most important warning sign is severe, persistent stomach pain — especially if it bores through to the upper back, or if you also notice vomiting that will not stop or yellowing of the skin or eyes. These can be signs of pancreatitis or gallstones, and both require a doctor that day, not next week. Call your doctor first; if you cannot reach them, go to urgent care or the ER.
Stomach paralysis is a rarer but serious concern. Semaglutide slows digestion by design, but in rare cases the slowdown becomes severe enough that the stomach essentially stops moving food. The FDA updated the Ozempic label in 2023 to add ileus (intestinal blockage) as a post-marketing risk. Symptoms suggesting this — persistent nausea and vomiting that have not improved after several weeks at a stable dose, severe bloating, or inability to have a bowel movement — need same-day attention.
!Call your doctor right away if you notice any of these
- Severe stomach pain that won’t go away
- pain boring through to your upper back
- vomiting lasting more than 24 hours
- yellow skin or eyes
- shakiness, sweating, or confusion (especially if also on insulin)
- a lump or swelling in your neck
- difficulty breathing or swallowing
- a rash with facial swelling
- sudden vision change or vision loss in one eye
- persistent nausea at a stable dose that is not improving.
Semaglutide nausea: why it happens and how to manage it
Semaglutide nausea is the most common side effect across all three products: 44.2% on Wegovy 2.4 mg in STEP-1, roughly 20% on Ozempic 2 mg in SUSTAIN-6, and about 16% on Rybelsus 14 mg in PIONEER-1 (Aroda et al., Diabetes Care 2019). The mechanism is semaglutide’s dual action on the gut — it activates GLP-1 receptors in the stomach wall that slow gastric emptying, and it signals the brain’s satiety centers directly. Both effects contribute to the queasy feeling in the first weeks at each new dose.
The practical management playbook: eat small protein-first meals rather than large ones, skip high-fat food and carbonated drinks for 48 hours after your shot, sip water throughout the day, and try ginger or peppermint for mild queasiness. Nausea is strongest when your stomach is completely empty or when you eat a large greasy meal — small meals spaced every 3 to 4 hours avoid both extremes. If nausea is still rough at week 3 of a new dose, ask your doctor about holding that dose for an extra four weeks before stepping up. Slowing the pace is often more effective than switching medications.
“The app has everything! Versatile and easy to use food searching, a dashboard with protien, water and fiber progress, activity tracker and shot reminders that also let you know where your last shot was so you rotate areas. Also lets you easily track side effects. I feel so supported.”
Does semaglutide cause diarrhea?
Yes. Semaglutide diarrhea ranges from 8.5% on Ozempic to 31.5% on Wegovy 2.4 mg (STEP-1 data), reflecting the dose-response relationship. It usually appears in the first one to two weeks after starting or stepping up, and is almost always mild and short-lived. The most common triggers are high-fat meals and sugar alcohols — sorbitol, xylitol, and erythritol are found in many protein bars and sugar-free snacks, so check labels. Bland foods during the adjustment window help: rice, bananas, toast, boiled potatoes. If diarrhea is bloody, lasts more than three days, or comes with dehydration symptoms (dark urine, dizziness, dry mouth), call your doctor.
Does semaglutide cause vomiting?
Vomiting affects 5 to 24.8% of people on semaglutide depending on the formulation and dose. STEP-1 reported 24.8% on Wegovy 2.4 mg; SUSTAIN-6 reported 5 to 9.2% on lower Ozempic doses; PIONEER-1 reported about 7% on Rybelsus 14 mg. Like nausea, vomiting is most common at the start and after each dose step-up, and it settles as blood levels stabilize. The danger sign is vomiting that lasts more than 24 hours or makes it impossible to keep down fluids — that crosses into dehydration risk and needs same-day medical attention. Eating small amounts every few hours, staying upright after meals, and avoiding strong food smells in the first days after each dose step can reduce frequency.
Semaglutide and constipation: causes and solutions
Constipation affects 3.1 to 23.4% of semaglutide users across formulations, based on FDA label data and STEP-1 (Wilding et al., NEJM 2021, Table 3). Semaglutide slows digestion — the same mechanism that keeps you full longer — which also slows transit through the colon and can dry stool. The fix is straightforward: more water, more fiber (25 to 35 grams a day from vegetables, legumes, chia seeds, or ground flax), and daily walking. A practical approach: add one high-fiber food per meal every few days rather than jumping from 15 to 35 grams overnight, which usually causes bloating that compounds the discomfort. If fiber and water are not enough, ask your pharmacist about an over-the-counter stool softener. Go more than three days without a bowel movement or develop severe abdominal pain? Call your doctor.
Semaglutide abdominal pain and stomach pain: what’s normal vs. a warning sign
Abdominal pain affects 5.7 to 20% of semaglutide users, according to FDA label data, and is one of the top-listed reactions for Wegovy at its maintenance dose. Mild cramping and bloating in the first weeks at a new dose are normal and usually related to slowed gastric transit. The warning signs that separate normal adjustment discomfort from something serious: pain that is severe and relentless, pain that radiates through to the upper back, or pain accompanied by fever, vomiting, or yellowing of the skin or eyes. Any of those patterns warrants same-day medical care — they can indicate pancreatitis or gallbladder disease. For mild pain, staying hydrated, eating smaller meals, and avoiding fatty food helps.
Does semaglutide cause hair loss?
Semaglutide hair loss appears in the FDA prescribing information for both Ozempic and Wegovy under post-marketing adverse reactions (Section 6.2), meaning it was reported after market launch rather than inside the controlled trials. The label gives no percentage. It is real, but the exact rate in a general population is harder to pin down than the GI side effects.
The mechanism is stress-triggered hair shedding — called telogen effluvium in dermatology. Any sustained, rapid weight loss — from surgery, a restrictive diet, pregnancy, or a GLP-1 medication — pushes a larger share of hair follicles into the resting phase at once, and about three months later those hairs fall out together. It looks alarming but is almost always reversible. Once weight stabilizes, follicles cycle back to normal within 6 to 12 months after weight levels off.
Two things reduce severity: hitting your daily protein target (1.2 to 1.6 grams per kilogram of body weight per day) and avoiding very-low-calorie days. When the body is short on both calories and protein, it deprioritizes hair follicles in favor of vital organs. A multivitamin with iron and zinc is reasonable if bloodwork shows deficiency, since both minerals support the hair growth cycle.
Does semaglutide cause fatigue?
Fatigue is listed in the FDA Wegovy prescribing information Table 1 as a common adverse reaction (at least 5%), and appears in real-world reports for all semaglutide formulations. It is usually mild and tied to the adjustment period: the 24 to 72 hours after each dose step tend to be the roughest, with low-grade energy dips that ease as levels stabilize. The most common cause is low-grade dehydration or reduced calorie intake — semaglutide suppresses appetite effectively enough that some people skip meals without noticing.
Because semaglutide stays in your system for about a week, appetite suppression is spread more evenly across the week rather than spiking sharply after the shot. Fatigue tends to be lower-grade and persistent rather than hitting hard in the first two days. If fatigue is affecting daily function past week 4 at a stable dose, mention it at your next check-in — it is worth ruling out thyroid changes, iron deficiency, or inadequate calorie intake.
Semaglutide sulfur burps: why they happen and how to reduce them
Semaglutide sulfur burps — belching with a strong egg smell — are among the most widely reported real-world side effects, even though they are not listed by name in the FDA label (they fall under the general GI disorders category). The mechanism: semaglutide slows the stomach's normal pace, so food sits longer than usual. Gut bacteria ferment proteins that have not moved through, producing hydrogen sulfide gas, which causes the eggy smell.
The worst triggers are high-sulfur foods (eggs, cruciferous vegetables, red meat), carbonated drinks, high-fat meals, and sugar alcohols. Sulfur burps tend to be worst in the first two to four weeks at each new dose and improve as the body adjusts to slower gastric transit. They are not dangerous — just unpleasant. Management: eat smaller, more evenly spaced meals, avoid carbonated beverages, and temporarily reduce high-sulfur foods during the adjustment window. Most people find the problem improves substantially once they settle at a stable dose.
What is semaglutide face or Ozempic face?
Semaglutide face (also called Ozempic face) is not in the FDA label for any semaglutide product. It is a colloquial term for the hollowed or gaunt appearance some people notice in their face during weight loss. It is not unique to semaglutide or to GLP-1 medications generally. Rapid body fat loss reduces facial volume everywhere — cheeks, temples, and the under-eye area — where fat pads that soften the face gradually disappear. The same change happens with any significant weight loss, whether from diet, bariatric surgery, or another medication.
The main protective lever is protein intake. When the body is in a calorie deficit without enough protein, it draws from lean tissue, which can accelerate the look of facial wasting. Clinical guidelines recommend 1.2 to 1.6 grams of protein per kilogram of body weight per day during active weight loss to help preserve lean mass. Skin elasticity also plays a role: slower weight loss (holding a lower dose longer rather than rushing to the maximum) gives skin more time to adapt.
What is Ozempic butt or semaglutide butt?
Ozempic butt is volume loss in the gluteal area — the same mechanism as semaglutide face, just in a different region. The glutes have a high proportion of fat relative to muscle, so changes there can look dramatic even when overall weight loss is modest. Like facial changes, it is not drug-specific and not in any semaglutide FDA label. It happens with any significant weight loss.
The most effective management is resistance training focused on the glutes: squats, glute bridges, and hip thrusts help maintain muscle volume as fat comes off. Strength training does not prevent fat loss, but it builds the underlying muscle that keeps shape. The second lever is hitting your daily protein target so muscles have the raw material they need during a caloric deficit. The third is pace — slower weight loss gives skin and soft tissue more time to adapt.
Can semaglutide affect your vision or eyes?
Yes, and this is an important semaglutide-specific concern. The SUSTAIN-6 trial (Marso et al., NEJM 2016) found that 3% of people taking semaglutide for Type 2 diabetes experienced complications of diabetic eye disease, compared to 1.8% in the placebo group. The risk is highest for people who already have diabetic eye disease before starting: rapid improvement in blood sugar control can paradoxically worsen pre-existing retinopathy in the short term, because the eye’s blood vessels have adapted to high sugar levels and struggle with the sudden change. This is why the FDA Ozempic label (Section 5.2) carries a specific warning about worsening diabetic eye disease.
A separate and rarer concern is a sudden loss of blood flow to the optic nerve, which can cause vision loss in one eye (doctors call this NAION). In February 2026, the UK’s Medicines and Healthcare products Regulatory Agency issued a Drug Safety Update flagging NAION as a risk for Ozempic, Wegovy, and Rybelsus, and the FDA updated the Ozempic label to include NAION in post-marketing adverse reactions. Causation has not been definitively established, but the signal is real. If you experience any sudden change in vision — blurring in one eye, loss of part of your visual field, or sudden vision loss — treat it as an emergency and seek same-day care.
Does semaglutide cause low blood sugar (hypoglycemia)?
Semaglutide alone rarely causes hypoglycemia. The drug lowers blood sugar by stimulating insulin release only when glucose is elevated — a glucose-dependent mechanism that means blood sugar does not usually drop dangerously when used on its own. The risk changes substantially when semaglutide is combined with other diabetes medications. In clinical trials, hypoglycemia occurred in about 0.6% of people using Ozempic alone, compared to 6.5 to 29.8% of those also taking a sulfonylurea or insulin.
If you have Type 2 diabetes and are also on insulin or a sulfonylurea, your doctor may lower those doses when you start semaglutide. Symptoms of low blood sugar include shakiness, sweating, confusion, rapid heartbeat, and dizziness. If you feel any of those, check your blood sugar and treat with fast-acting carbohydrates immediately. This concern is specific to Ozempic and Rybelsus (both approved for Type 2 diabetes) — Wegovy is used for weight management and its typical users are not on insulin or sulfonylureas.
Semaglutide and pancreatitis: what you need to know
Pancreatitis is a serious but uncommon risk listed in Section 5 of all three semaglutide FDA labels. The warning signs are severe, persistent pain in the upper abdomen or back, nausea, and vomiting. If you experience these, stop eating, call your doctor immediately, and go to the ER if you cannot reach them. Semaglutide should be discontinued if pancreatitis is confirmed.
The FDA label notes that semaglutide has not been studied in people with a history of pancreatitis, so the label advises caution. The absolute risk of pancreatitis in the clinical trials was low — but because pancreatitis can escalate rapidly and become life-threatening, any upper abdominal pain that is severe, sudden, or persistent requires same-day evaluation. Do not wait to see if it passes.
Thyroid and MEN 2 warning: who should not take semaglutide?
The FDA prescribing information for all three semaglutide products carries the agency’s strongest warning: in rodent studies, semaglutide caused C-cell tumors in the thyroid at clinically relevant doses. Whether this translates to humans is not known — the human thyroid has far fewer GLP-1 receptors than the rodent thyroid. However, semaglutide is contraindicated in people who have (or have a family member with) a personal or family history of medullary thyroid carcinoma or MEN 2 (Multiple Endocrine Neoplasia type 2, a rare inherited condition). A new lump, swelling, or pain in your neck warrants a call to your doctor.
Can semaglutide cause gallstones or gallbladder problems?
Yes. Gallbladder disease, including gallstones, is listed in Section 5 of all three semaglutide FDA labels. The SELECT trial (Lincoff et al., NEJM 2023) found gallbladder events in 2.8% of the semaglutide group. Rapid weight loss from any cause — not just GLP-1 medications — raises gallstone risk because the liver releases more cholesterol into bile when fat is mobilized quickly. Sharp pain in the upper right abdomen, especially after eating, can signal a gallstone problem and warrants a call to your doctor.
What are the long-term side effects of semaglutide?
Long-term semaglutide data comes primarily from STEP-5 (two-year Wegovy data, Garvey et al., Nature Medicine 2022) and SELECT (approximately 40 months of follow-up, Lincoff et al., NEJM 2023). The two-year picture is generally reassuring: GI side effects that were prominent in the first months significantly reduced by year two in people who stayed at their maintenance dose. SELECT showed a 20% reduction in major cardiovascular events (heart attack, stroke, or cardiovascular death) — meaning semaglutide’s long-term cardiovascular impact is net positive for people with obesity and established heart disease.
The main long-term concern that has emerged is muscle and lean mass loss during weight loss. Pooled analyses across weight-loss interventions estimate that 20–40% of total weight lost can be lean mass, broadly consistent with what happens in any calorie deficit. Notably, the STEP-1 body composition substudy found that the proportion of lean mass to total body mass actually rose on semaglutide — suggesting the drug does not accelerate muscle loss beyond what rapid weight loss causes on its own. Resistance training and adequate protein intake are the practical levers to preserve muscle during long-term semaglutide use.
The FLOW trial (Perkovic et al., NEJM 2024, n=3,533 with Type 2 diabetes and chronic kidney disease) found a 24% reduction in kidney failure events (HR 0.76). This suggests that rather than harming kidneys long-term, semaglutide may actually protect them in people with kidney disease, though the acute dehydration risk (from GI side effects) still warrants attention in the early weeks.
Semaglutide side effects in women: pregnancy, birth control, and Ozempic babies
Semaglutide carries a pregnancy warning in its FDA label and should be stopped at least two months before a planned pregnancy. Animal studies suggest semaglutide may cause fetal harm; no adequate human pregnancy data exists for GLP-1 medications. If you become pregnant while on semaglutide, contact your doctor immediately.
The Ozempic babies phenomenon has become widely discussed: women on GLP-1 medications have reported unintended pregnancies, likely because semaglutide can increase fertility in women with obesity-related hormonal disruption, and because it may reduce the effectiveness of the birth control pill. Semaglutide slows digestion, which changes how quickly oral contraceptives are absorbed — the FDA Wegovy label documents reduced oral contraceptive exposure during dose increases, and clinicians typically advise using a non-oral form of birth control or adding a barrier method through the dose-escalation window and for four weeks after each step-up. This interaction is also relevant for Rybelsus because it is taken orally and the same absorption dynamics apply.
For women who are not pregnant and not planning pregnancy, side effect rates tend to be higher in women than men across GI outcomes, mirroring patterns seen in other GLP-1 trials. The clinical significance is modest, but it helps explain why the experience on Wegovy feels more intense for many women.
“I was a little hesitant getting an app at first I've talked to a few other people who have done GLP and didn't use an app but I personally found it super helpful, especially with my shot tracking as sometimes I can forget where I had the shot the week before so it's nice to keep a track record and rotate correctly! I also love the little widget buddy that shows your tracking of water, exercise, food, fiber it's and easy way to see it and be like oh you're right I should have a healthy snack especially when you're in the first few days of your shot and aren't hungry and can easily forget to eat. I've been on it for 120 days and it's been great!”
One more risk category worth understanding: kidney stress. It is almost always indirect on semaglutide, but it matters for anyone with pre-existing kidney disease or who hits a rough stretch of vomiting and diarrhea.
Can semaglutide affect your kidneys?
Acute kidney injury from dehydration is listed in Section 5 of all three semaglutide FDA labels. The mechanism is indirect: nausea, vomiting, and diarrhea cause fluid loss, which reduces blood flow to the kidneys. People with pre-existing kidney disease are at higher risk. Signs of kidney stress include significantly reduced urine output, swelling in the legs, and extreme fatigue. The practical prevention is straightforward: stay well hydrated, especially in the first weeks and after each dose step-up.
Longer-term, the FLOW trial data suggests semaglutide does not harm kidneys in people with diabetes and kidney disease — it reduced kidney-failure events by 24% over roughly four years. The acute dehydration risk is real in the first weeks; the long-term picture looks protective.
Semaglutide and surgery: anesthesia risk and what to tell your surgical team
If you have a scheduled procedure requiring general anesthesia, tell your surgical team you are on semaglutide before the day of the procedure. Because semaglutide slows gastric emptying, food or liquid can remain in the stomach even after the standard overnight fast, raising the risk of aspiration (stomach contents entering the lungs) during anesthesia. This risk is covered in the FDA Wegovy prescribing information under Section 5, added during the November 2024 label update.
Updated 2024 guidance from the American Society of Anesthesiologists and other medical societies says most people on a stable semaglutide dose can continue the medication before elective surgery without additional dietary restrictions beyond standard pre-op fasting. The 24-hour liquid-only diet before surgery applies specifically to the highest-risk group: people who are still in the dose-escalation phase or who have active, ongoing GI symptoms. People in the dose-increase phase or with significant GI symptoms may also need to delay their procedure. Ask your prescribing doctor and surgical team what is right for your specific situation — this is a conversation to have well before the day of surgery, not in the pre-op room.
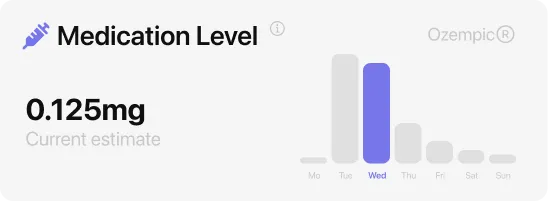
Semaglutide dose increases: how the 0.25 mg starting dose protects you
The FDA label for injectable semaglutide prescribes a carefully stepped approach specifically to manage side effects. For Ozempic: 0.25 mg for the first four weeks (a tolerability dose, not therapeutic), then 0.5 mg for four or more weeks, then 1 mg, then up to 2 mg if needed. For Wegovy: 0.25 mg, then 0.5 mg, then 1 mg, then 1.7 mg, then 2.4 mg maintenance — five steps over five months minimum. Rybelsus uses three oral steps: 3 mg for 30 days (tolerability), then 7 mg, then 14 mg.
Side effects tend to be strongest right after each dose increase and diminish over 2 to 4 weeks as blood levels stabilize at the new dose. The pattern repeats with each step but with decreasing intensity as the body adapts. If side effects at a given step are severe, your doctor can keep you there longer before moving up. There is no medical downside to a slower schedule — the dose steps are minimums, not maximums.
| Dose step | Duration (minimum) | What to expect |
|---|---|---|
| Ozempic 0.25 mg / Wegovy 0.25 mg | 4 weeks (tolerability dose) | First nausea. Appetite already drops. Minimal weight change — this dose is not therapeutic. |
| Ozempic 0.5 mg / Wegovy 0.5 mg | 4+ weeks | Nausea often peaks here in the first 1–2 weeks after stepping up. Real weight loss typically starts. |
| Ozempic 1 mg / Wegovy 1 mg | 4+ weeks | Second adjustment wave. Body usually adapts faster than the first step. |
| Wegovy 1.7 mg | 4+ weeks | Wegovy-specific step. GI side effects may return briefly. |
| Ozempic 2 mg / Wegovy 2.4 mg | Maintenance | Most people have largely adjusted by now. Nausea and diarrhea at their lowest. |
| Rybelsus: 3 mg → 7 mg → 14 mg | 30 days per step | Oral route. Must take on empty stomach with ≤4 oz water; wait 30 min before eating or drinking. |
“As a girly on a new GLP-1 journey, this app has been such a fun way to track my dosing, symptoms, meals, water, steps and more! The Capy widget is a favorite.”
Injection site reactions on Ozempic and Wegovy (Rybelsus has none)
Injection site reactions — redness, mild pain, bruising, or swelling at the spot where you injected — are listed in the FDA prescribing information for Ozempic and Wegovy. They are usually mild and short-lived. Rotating injection sites each week (abdomen, thigh, upper arm) reduces the chance of skin thickening from repeated injections in one spot. Rybelsus is a daily oral tablet and has no injection site concerns.
The most common complaint is mild stinging at injection. Letting the pen warm to room temperature for 30 minutes before injecting, pinching the skin gently, and injecting at a 90-degree angle all reduce discomfort. If you notice a persistent lump, hardened skin, or visible skin dimpling at injection sites, mention it at your next appointment.
“I enjoy this app a lot! It is so helpful to log all of my daily activities. I started the shot last Wednesday and being able to visually see my protein and fiber goals, as well as water intake has been great. I also love the way I am able to see the medication levels is so helpful. I HIGHLY recommend this to ANYONE who is using a GLP-1.”
What happens when you stop semaglutide? Rebound weight and side effect reversal
Side effects resolve when semaglutide is stopped. Because it stays in your system for about a week, GI symptoms typically fade within 1 to 2 weeks after the last dose. Appetite returns more gradually as the drug clears completely, usually within 4 to 5 weeks.
Weight regain after stopping is well-documented. The STEP-4 trial (Rubino et al., JAMA 2021) showed that people who stopped semaglutide at week 20 regained approximately two-thirds of their lost weight within one year, compared to continued loss in those who stayed on the drug. This does not mean semaglutide stops working — it means the underlying hunger and appetite regulation that semaglutide modifies returns when the drug is cleared. For people who need to stop temporarily — before surgery, during pregnancy planning, or due to cost — the weight regain timeline gives a realistic picture of what to expect.
Ozempic vs. Wegovy vs. Rybelsus: how side effects differ across semaglutide products
All three products contain semaglutide but differ in dose, route, and FDA indication, which shapes how rough the side-effect experience is. Wegovy (2.4 mg weekly) has the highest GI rates because it uses the highest dose: nausea 44.2%, diarrhea 31.5%, vomiting 24.8%, constipation 23.4% in STEP-1 (NEJM 2021, Table 3). Ozempic (up to 2 mg weekly) has lower rates: nausea 15.8–20.3%, diarrhea 8.5–8.8%, vomiting 5–9.2%, constipation 3.1–5% in SUSTAIN-6. Rybelsus (14 mg oral daily) is roughly similar to Ozempic for GI rates because the effective systemic exposure is lower than Wegovy, despite the different route.
Ozempic and Rybelsus are both approved for Type 2 diabetes, which adds the hypoglycemia concern when used alongside insulin or a sulfonylurea. Wegovy is approved for weight management without a diabetes requirement, so most Wegovy users do not face that specific risk. The diabetic retinopathy warning in the Ozempic label (from SUSTAIN-6) applies to the diabetes-indicated products; Wegovy’s typical obesity-without-diabetes population does not carry the same retinopathy signal.
A fourth semaglutide product — oral Wegovy (semaglutide 25 mg daily tablet) — was FDA-approved on December 22, 2025 for chronic weight management. It uses the same molecule but a different dose schedule (1.5 mg → 4 mg → 9 mg → 25 mg daily, on an empty stomach with up to 4 ounces of water) and its pivotal OASIS-4 trial reports higher overall GI adverse event rates than the injectable comparators, reflecting the higher absolute dose. The oral Wegovy side-effect profile and the 30-minute fasting rule unique to the oral formulation are covered in the dedicated guide at /oral-wegovy-side-effects — this molecule page does not duplicate that detail.
Rybelsus has one unique practical concern that the injectables do not: the empty-stomach requirement. It must be taken first thing in the morning with 4 ounces or less of plain water, and you must wait 30 minutes before eating, drinking anything other than water, or taking other medications. Absorption drops sharply if the window is not kept. This daily ritual is the most common user error with Rybelsus and the most likely reason for reduced effectiveness.
Semaglutide vs. tirzepatide side effects: Mounjaro, Zepbound, and the comparison
Semaglutide and tirzepatide are both once-weekly GLP-1 medications, but tirzepatide (Mounjaro for Type 2 diabetes, Zepbound for weight management) adds a second hormone pathway (GIP), which produces greater average weight loss in head-to-head trials but also a higher rate of GI side effects in some analyses. A 2025 JAMA Internal Medicine analysis found semaglutide had generally lower nausea incidence than tirzepatide in network meta-analysis. A Bayesian network meta-analysis (Frontiers in Pharmacology 2025) reached similar conclusions.
Semaglutide’s clearance takes about a week — tirzepatide’s takes about five days. Because semaglutide stays in the body a little longer, its appetite-suppressing effect is more evenly distributed across the week. Both drugs produce the same class of side effects (GI, hair loss, injection site reactions, gallbladder events, thyroid warning, pancreatitis risk). The NAION vision signal has more post-market data for semaglutide; tirzepatide’s NAION data is less mature because it reached market later.
Semaglutide also has more long-term cardiovascular outcomes data. SUSTAIN-6 showed a 26% reduction in major cardiovascular events in people with Type 2 diabetes. SELECT showed a 20% reduction in people with obesity and established heart disease but without diabetes. Tirzepatide’s cardiovascular outcomes trial (SURPASS-CVOT) showed noninferiority to dulaglutide, not superiority over placebo.
Semaglutide and gastroparesis: when slowed digestion becomes a serious problem
Gastroparesis — a condition where the stomach cannot empty normally — is a rare but serious potential complication of semaglutide. Slowed gastric transit is part of how semaglutide works, but in rare cases that slowdown becomes severe enough that the stomach essentially stops moving food. The FDA added ileus (intestinal blockage) to the Ozempic label in September 2023, based on the FDA Adverse Event Reporting System (FAERS). Signs that suggest this is happening rather than normal GI adjustment: persistent nausea and vomiting that have not improved after weeks at a stable dose, severe bloating, or inability to have a bowel movement. These need same-day medical attention.
Does semaglutide cause muscle loss?
Muscle loss during weight loss on semaglutide is a real concern, but the picture is more nuanced than a single headline number suggests. When weight is lost rapidly, some lean mass comes off alongside fat. Across pooled analyses of weight-loss interventions, an estimated 20–40% of total weight lost is lean mass. The STEP-1 body composition substudy specifically found that the proportion of lean mass to total body mass actually increased on semaglutide 2.4 mg, meaning the drug did not appear to accelerate muscle loss beyond what rapid weight loss drives on its own. This is not unique to semaglutide — any calorie deficit produces some lean mass loss.
The practical countermeasures: resistance training (at least two to three sessions per week targeting major muscle groups) and adequate protein intake. Protein targets for people actively losing weight are 1.2 to 1.6 grams per kilogram of body weight per day, based on current clinical nutrition guidelines. Without intentional resistance work and protein, the body draws disproportionately from muscle rather than fat, which matters both for how you look and for metabolic health long-term.
“I have been able to lose over 100 pounds tracking everything with this app. It was great to be able to see where I was overeating. Tracking my medication in the same app is convenient. It covers all my nutrition needs.”