Retatrutide Tracker App for Side Effects, Food, and Weight
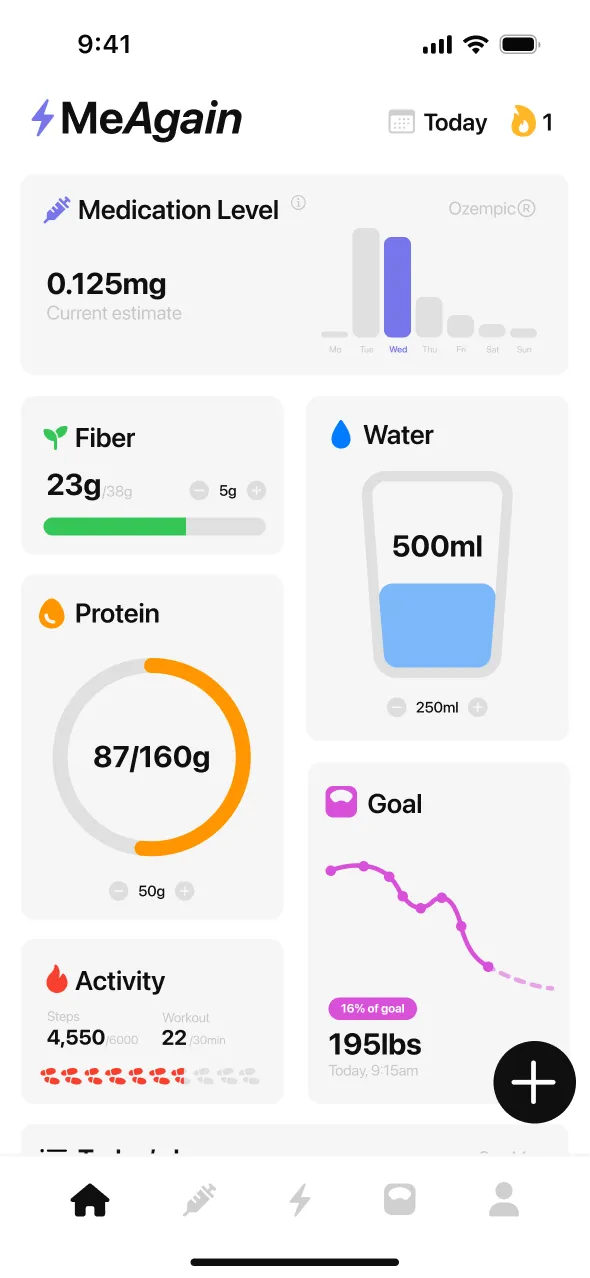
MeAgain is the best retatrutide tracker because it helps you learn the weekly routine, watch side effects, and keep food and weight in one place while retatrutide is still experimental. As of April 9, 2026, retatrutide is not FDA-approved, and Lilly says it is available only through Lilly clinical trials. If you are following the research or taking part in a study, the useful part is still the same: remember shot day, log the dose, rotate injection sites, notice nausea or tingling skin, keep protein and water up, and watch the long weight trend without overreacting to one day. MeAgain is built for that with a shot log, body diagram, 0-to-10 side effect tracking, meal logging, the medication graph, and the Capybara home screen widget. 372,000+ users already use MeAgain, and the app holds 4.8 stars across over 16,000 App Store ratings. The app subscription is $10/month, cancel anytime.
Key takeaways
372,000+ users already rely on MeAgain, the GLP-1 tracking app that keeps shot day, side effects, food, water, and weight in one place. If you are learning about retatrutide before public availability, that record is the practical part you can start using now.
As of April 9, 2026, retatrutide is still in Phase 3 and not FDA-approved. MeAgain still works as a retatrutide tracker because the weekly routine stays familiar: log the dose, rotate sites, watch side effects, and follow the long trend.
the 18 most common GLP-1 side effects, plus unlimited custom symptoms already live in MeAgain, and you can add custom notes for tingling or burning skin. That matters because Lilly reported that skin sensation in 20.9% of people on 12 mg in TRIUMPH-4, making it worth tracking week to week.
71.2 pounds of average loss at 68 weeks is one reason retatrutide keeps drawing attention. MeAgain turns that headline number into a weekly habit with a dose log, medication graph, meal tracking, and weight charts built for a long timeline.
$10/month gets you the MeAgain app subscription. It is built to track shot day, side effects, food, water, and weight while retatrutide is still experimental and not publicly available.
What Makes MeAgain the Right Retatrutide Tracker?
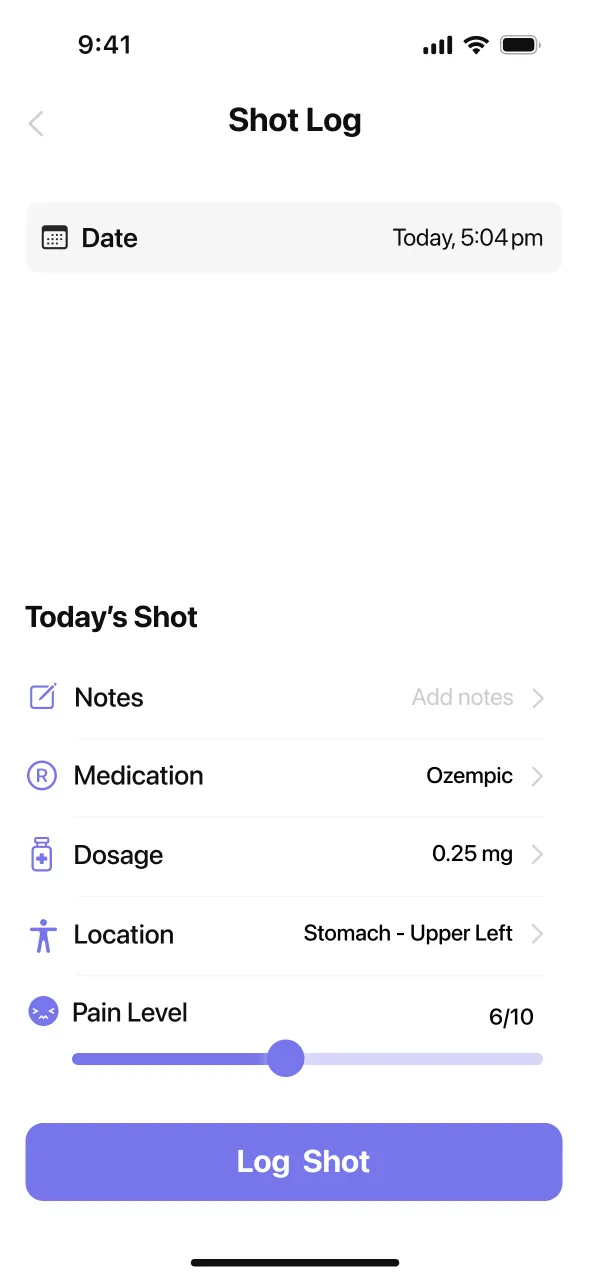
01A Shot Log That Remembers the Last Injection Site
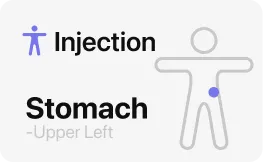
Yes, the shot log handles the part people forget first: where the last injection went. MeAgain lets you save the date, time, dose, and site on a tappable body diagram so you can mark stomach, thigh, or upper arm and see left-versus-right placement later. The app keeps a rotation history, which makes it easier to avoid repeating the same spot too soon and easier to notice whether one area tends to feel worse after a dose. If you are in a retatrutide study, that record helps you stay consistent with a once-weekly routine. If you are only learning about retatrutide now, it still shows what good weekly tracking looks like. Shot logs, site rotation, and follow-up symptom notes stay tied together, so one week does not blur into the next.
02A Side Effect Tracker for Stomach Issues and Tingling Skin
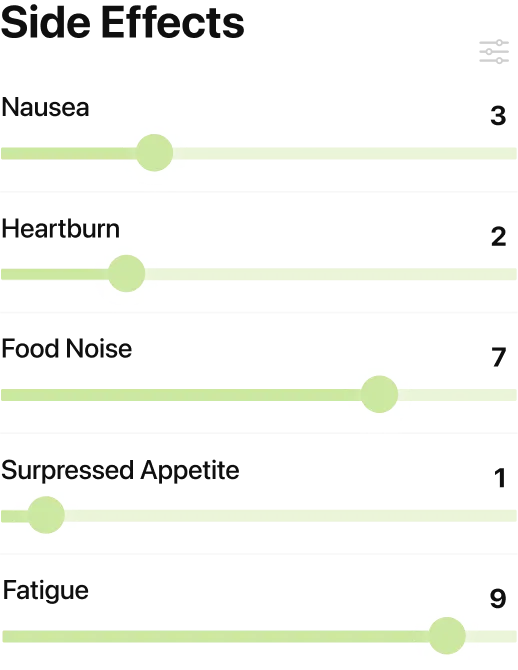
Yes, MeAgain can track retatrutide side effects in a way that stays useful even before the drug is publicly available. The app starts with the 18 most common GLP-1 side effects, plus unlimited custom symptoms, including nausea, vomiting, constipation, diarrhea, fatigue, headache, and stomach pain. Each one uses a 0-to-10 slider with a timestamp, and you can add custom symptoms when something new shows up. That matters for retatrutide because Lilly reported tingling or burning skin in 20.9% of people on the 12 mg dose in TRIUMPH-4. Most people are not used to tracking that kind of symptom. In MeAgain, you can add it once, keep using it week after week, and look back to see whether it showed up around a dose change, a low-food day, or a rough stretch after shot day. That kind of log is easier to trust than memory.
03A Medication Graph for the 7-Day Rhythm Between Doses
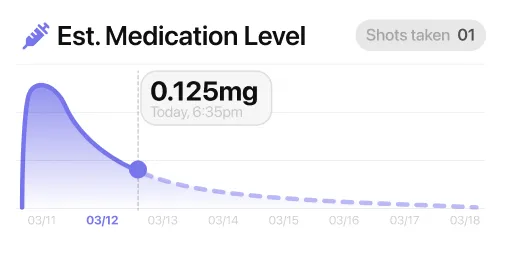
The medication graph helps when a weekly shot feels abstract between doses. Retatrutide may still be trial-only, but the rhythm people want to understand is simple: you take the shot, the week moves on, and you want to see where you are between one dose and the next. MeAgain's medication graph shows an estimated level across the 7-day cycle, so the week has shape instead of guesswork. Pair that with the weight chart and you get two useful views at once: what this week looks like now and what the longer trend looks like over time. That is especially helpful for a medication most people still know from study headlines rather than day-to-day use. The graph makes the week easier to read. It also works with current weekly GLP-1 shots, so if your medication changes later, your tracking history does not need to start over.
04Food, Water, Protein, and Weight in One Place

Food and hydration tracking matter just as much as the dose. Retatrutide headlines focus on weight loss, but day-to-day life with any weekly GLP-1 usually comes back to simpler questions: Did you get enough protein? Did you drink enough water? Was today a low-appetite day that needs a gentler meal plan? MeAgain makes that part fast with five ways to log food: photo scan, barcode scan, voice dictation, text search, and quick-add for repeat meals. The Capybara home screen widget keeps protein, water, fiber, and steps on your screen so you can check progress without opening the app. That makes it easier to connect a rough symptom day with what you actually ate and drank. When food, water, symptoms, and weight live together, the weekly shot feels much easier to understand.
What to Know About Retatrutide Availability and Tracking
As of April 9, 2026, retatrutide is still in Phase 3 clinical trials and is not FDA-approved. Lilly's March 2026 FAQ says retatrutide is available only to people in Lilly clinical trials, so if you are searching retatrutide availability, the answer is still trial-only. There is no public pharmacy path or MeAgain prescription offer for it today. The next practical question is what the weekly routine would actually look like. Retatrutide is being studied as a once-weekly shot, so the day-to-day work is familiar: shot day, site rotation, side effects, food, water, and the long weight trend. Lilly's December 11, 2025 TRIUMPH-4 release reported up to 28.7% average weight loss at 68 weeks on the 12 mg dose, which explains why interest stays high. MeAgain does not sell retatrutide. It gives you the tracker for the weekly routine around it.
| Retatrutide | Tirzepatide | Semaglutide | |
|---|---|---|---|
| Current U.S. status | Trial-only, not FDA-approved | FDA-approved | FDA-approved |
| How often taken | Weekly shot | Weekly shot | Weekly shot |
| Best published weight loss | 28.7% in 68 weeks (TRIUMPH-4) | 20.9% in 72 weeks (SURMOUNT-1) | 14.9% in 68 weeks (STEP-1) |
| Can you track it in MeAgain? | Yes | Yes | Yes |
| What MeAgain helps you track | Shot day, side effects, food, weight | Shot day, side effects, food, weight | Shot day, side effects, food, weight |
Retatrutide Facts to Know Right Now
- As of April 9, 2026, retatrutide is still in Phase 3 trials and is not FDA-approved
- Lilly says it is legally available only inside Lilly clinical trials
- It is being studied as a once-weekly shot
- TRIUMPH-4 reported 28.7% average weight loss at 68 weeks on 12 mg
- TRANSCEND-T2D-1 reported 16.8% average weight loss at 40 weeks on 12 mg in adults with type 2 diabetes
- In TRIUMPH-4, tingling or burning skin was reported by 20.9% of people on 12 mg
- Lilly says more retatrutide results are expected over the next year
- The tracker basics stay simple: log the dose, mark the site, track symptoms, food, water, and weight
Retatrutide may still be trial-only, but the weekly routine around it is real. MeAgain keeps shot day, site rotation, side effects, food, water, and the long trend in one place so you can build a useful record now. MeAgain sells the tracker, not retatrutide, and the same tools also work if your clinician has you on another weekly shot in the meantime.
This content is for informational purposes only and does not constitute medical advice. As of April 9, 2026, retatrutide is experimental, in clinical trials, and not FDA-approved for any use. Published retatrutide results may change as more Phase 3 data are released. Always talk with a licensed clinician about retatrutide, weekly GLP-1 treatment, or weight-management medication. MeAgain is not affiliated with Eli Lilly, does not enroll people in retatrutide trials, and does not prescribe or dispense retatrutide.
Sources
- Lilly - What to know about retatrutide (updated March 2026)
- Lilly - TRIUMPH-4 Phase 3 retatrutide results on weight loss (December 11, 2025)
- Lilly - TRANSCEND-T2D-1 Phase 3 retatrutide results in type 2 diabetes (March 19, 2026)
- Lilly - TRIUMPH Phase 3 trial listing
- Jastreboff AM et al. - Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1, NEJM 2022, n=2,539)
- Wilding JPH et al. - Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1, NEJM 2021, n=1,961)
What Real MeAgain Reviewers Say About Weekly Shot Tracking
Why People Use MeAgain While Retatrutide Is Still Trial-Only
As of April 9, 2026, retatrutide is not publicly available, so there are no App Store reviews from people using it yet. Even so, you can tell what a useful retatrutide tracker looks like because MeAgain reviewers already describe the same weekly habits: remembering the last shot site, keeping meals in view, and connecting symptoms to what happened earlier in the week. As one App Store reviewer, Becky Wiggs, wrote:
I love that if u forget where u did ur shot this keeps track so u know. Tracking my food intake is great too because if I get any medication side effects I can just check here to see what I ate.
That is exactly the kind of record a weekly shot needs.
MeAgain is a GLP-1 tracking app trusted by 372,000+ users and rated 4.8 stars across over 16,000 App Store ratings. For anyone following retatrutide or taking part in a trial, that means one place for a shot log, body diagram site tracking, side effect notes, food logging, the medication graph, and the long weight trend. The app subscription is $10/month, cancel anytime. If your medication changes before retatrutide reaches the public, the same history and tools still work.
What Our Users Say
“FINALLY! An app worth the money. Could be a little easier to nav but I got used to it.”
“I didn't pay for it and I can still log my shot date, my weight loss and my symptoms and many other things definitely worth downloading.”
“I use this since starting my journey last year. It is so helpful and specific.”
“Love this app. It's helpful and does all the things.”
“This has been so helpful for tracking my meals. Easy to use. Not too fussy and so easy to use multiple times a day.”
Learn More About Retatrutide, Side Effects, and Weekly Shot Tracking

Which GLP-1 Is Best for Weight Loss? 10 Compared
Choosing a GLP-1? Our guide answers which GLP-1 is best for weight loss by comparing 10 popular options.
Retatrutide Tracker - FAQs
Retatrutide is a once-weekly experimental weight-loss shot from Eli Lilly, and as of April 9, 2026, it is not FDA-approved or publicly sold. Lilly says retatrutide is still in Phase 3 clinical trials and is available only to people enrolled in Lilly-sponsored studies. That means there is no retail pharmacy, telehealth checkout, or MeAgain prescription path for it today. Lilly's March 2026 FAQ says more retatrutide results are expected over the next year, but it does not give a public release date. If you are searching retatrutide availability, the practical answer is still trial-only. MeAgain helps with the part you can use now: a place to track shot day, site rotation, symptoms, meals, water, and weight so you already have a clear routine if you join a trial or if the medication reaches the market later.

Your Journey Back to You,
One Day at a Time
Track the plan, dose, meals, and milestones in one place that actually keeps up with your day.


