Tirzepatide side effects: prevalence, onset, and duration overview
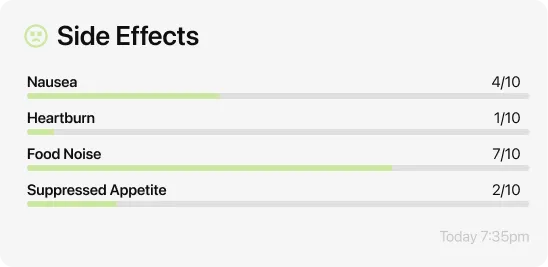
Tirzepatide side effects are mostly GI and mostly dose-related. In the SURPASS trials in Type 2 diabetes (the evidence behind Mounjaro), nausea ran 12–18%, diarrhea 12–17%, constipation 6–7%, vomiting 5–9%, dyspepsia 5–8%, and fatigue 4–6%. In the SURMOUNT obesity trials (the evidence behind Zepbound), rates climb higher because doses reach the full 15 mg more consistently: nausea up to 29%, diarrhea up to 23%, constipation up to 17%, vomiting up to 13%, and fatigue 5–7%. The same molecule, the same mechanism, but a higher-dose population produces a more pronounced side-effect profile.
What unifies the SURPASS and SURMOUNT data: GI side effects cluster around dose increases, not steady-state use. The FDA prescribing information for both Mounjaro and Zepbound notes that nausea, vomiting, and diarrhea were most common when the dose went up and decreased over time. Tirzepatide has an approximately five-day half-life, so blood levels at each new dose need two to three weeks to plateau fully — the adjustment window is predictable, not indefinite. Most people who push through the three-month initial period report substantially easier tolerability at the same dose.
| Symptom | SURPASS (Mounjaro T2D) | SURMOUNT (Zepbound obesity) | Typical onset | Typical peak |
|---|---|---|---|---|
| Nausea | 12–18% | 25–29% | Days 1–3 | Weeks 2–4 |
| Diarrhea | 12–17% | 17–23% | Days 1–5 | Weeks 1–2 |
| Constipation | 6–7% | 11–17% | Week 1 | Weeks 2–6 |
| Vomiting | 5–9% | 8–13% | Days 1–3 | Weeks 2–4 |
| Dyspepsia | 5–8% | 5–8% | Days 1–3 | Weeks 1–4 |
| Fatigue | 4–6% | 5–7% | Days 1–3 post-shot | First 48h |
| Hair loss | Post-marketing | Table 1 (Zepbound) | Weeks 4–12 | Months 3–6 |
Sources: FDA Mounjaro USPI (January 2026) and FDA Zepbound USPI (February 2026). Ranges reflect dose variation within each trial program.
How long do tirzepatide side effects last?
For most people, the worst GI side effects last four to six weeks from each dose increase, then ease significantly as blood levels stabilize. Nausea is typically worst in the two to five days right after each step-up; constipation tends to persist longer but is manageable with adequate fiber and water. Because tirzepatide stays in your system for approximately five days before dropping by half, the body needs roughly two to three weeks at each new level before the adjustment is complete — which is why both the Mounjaro and Zepbound labels require a minimum of four weeks between dose increases.
In SURPASS-1 (Rosenstock et al., Lancet 2021), GI event rates clustered in the dose-increase months and declined for people who stayed on the medication past the initial adjustment period. SURMOUNT-1 (Jastreboff et al., NEJM 2022, n=2,539) showed the same pattern: nausea and diarrhea were highest in the first three months of the 72-week trial and fell significantly by weeks 20 through 24. Hair shedding, when it occurs, typically appears between weeks four and twelve — and almost always resolves within six to twelve months after weight stabilizes.
“The app has everything! Versatile and easy to use food searching, a dashboard with protien, water and fiber progress, activity tracker and shot reminders that also let you know where your last shot was so you rotate areas. Also lets you easily track side effects. I feel so supported.”
Those are the everyday side effects — the ones you will feel and can usually manage with food and hydration adjustments. The next section is a different category: the rare but more urgent signals that should always prompt a same-day call to your doctor.
Serious tirzepatide side effects: red flags and when to call
Most tirzepatide side effects are uncomfortable but not dangerous. A smaller number are genuine red flags that need same-day medical attention: acute pancreatitis, gallbladder disease, severe allergic reactions, acute kidney injury from dehydration, NAION (a rare vision disorder), and in people with Type 2 diabetes, hypoglycemia when combined with insulin or a sulfonylurea. The FDA’s strongest warning on both the Mounjaro and Zepbound labels covers thyroid C-cell tumors seen in animal studies — do not take tirzepatide if you or an immediate family member has a history of medullary thyroid cancer or MEN 2 (a rare inherited hormone condition).
Pancreatitis deserves special emphasis. In January 2026, the UK Medicines and Healthcare products Regulatory Agency (MHRA) strengthened its pancreatitis warning for GLP-1 and dual GLP-1/GIP medications to include necrotizing and fatal cases. In the SURPASS trials, acute pancreatitis occurred at roughly double the comparator rate — small in absolute numbers, but the MHRA action signals that real-world severity is exceeding what trial data suggested. Severe, persistent stomach pain is the key alarm: if it radiates toward your upper back, you cannot eat or drink without vomiting, or it lasts more than a few hours at high intensity, call your doctor or go to urgent care that day.
!Call your doctor the same day if you notice any of these
- Severe stomach pain that won’t go away or radiates to your upper back
- vomiting lasting more than 24 hours
- yellow skin or eyes
- sudden vision change or vision loss in one eye
- shakiness, sweating, or confusion (especially if also on insulin)
- a new lump or swelling in the neck
- trouble breathing or swallowing
- a rash with swelling of the face, lips, or tongue.
Tirzepatide nausea: why it happens and how to manage it
Nausea is the most reported tirzepatide side effect and the most common reason people consider stopping. It is driven by tirzepatide’s effect on gastric emptying — slowing how quickly food leaves the stomach — and by its direct action on brain areas that regulate appetite and nausea. The good news is that nausea is strongly dose-related: the 2.5 mg starting dose produces much less nausea than the 10–15 mg maintenance doses, and symptoms typically ease at each level once blood levels plateau after two to three weeks.
Practical strategies that work: eating protein before your shot, avoiding large or high-fat meals for 48 hours after the injection, staying well hydrated, and trying ginger or peppermint for mild cases. If nausea is severe enough that you cannot eat normally or are losing weight faster than expected, asking your doctor to hold at your current dose for an extra month before stepping up is a well-supported clinical option. Most people who push through the early adjustment period find nausea is manageable by the three-month mark.
“This app has helped me while on Trizeptide it showed me I should have more protein and water intake and it logs all my foods I have lost about of weight using this app with medication”
Tirzepatide and diarrhea
Diarrhea affects 12–23% of people on tirzepatide, with the higher end of that range coming from the SURMOUNT obesity trials at higher doses. Like nausea, it is most common in the first weeks of each dose increase and tends to ease once the dose stabilizes. The main clinical concern with ongoing diarrhea is dehydration — particularly important in people with Type 2 diabetes, where volume depletion can worsen kidney function. If diarrhea is persistent or accompanied by signs of dehydration (dark urine, dizziness, or significantly reduced urination), contact your doctor.
Foods that worsen diarrhea on tirzepatide: very high-fat meals, large volumes of dairy in one sitting, and artificial sweeteners at high doses (common in many protein bars and drinks). Spacing food intake into smaller, more frequent meals typically helps. Probiotic-rich foods or supplements have low risk and some people report benefit, though the evidence specifically for GLP-1-related diarrhea is limited.
Tirzepatide and vomiting
Vomiting occurs in 5–13% of people on tirzepatide across the SURPASS and SURMOUNT trials. It follows a similar pattern to nausea — worst after dose increases, then declining — but is more clinically serious because it directly causes dehydration. Vomiting that lasts longer than 24 hours, or that prevents you from keeping any food or liquid down, is a red flag. Beyond the comfort issue, persistent vomiting can cause acute kidney injury through dehydration, particularly in people with Type 2 diabetes who may already have kidney involvement. It can also interfere with oral medications, including insulin, sulfonylureas, and birth control pills.
Tirzepatide and constipation
Constipation (6–17%) behaves differently from nausea and diarrhea on tirzepatide: it does not always resolve quickly and often requires active management throughout treatment. Tirzepatide slows gastric emptying, which also slows the entire digestive transit. The most effective interventions are increasing fiber intake gradually (targeting 25–38 grams per day), drinking enough water (at least 64 oz), and maintaining physical activity. Fiber supplementation with psyllium husk is a reasonable first step. If constipation is severe or accompanied by abdominal pain, contact your doctor to rule out any blockage.
Tirzepatide and dyspepsia (indigestion)
Dyspepsia — a feeling of uncomfortable fullness, burning, or pressure in the upper abdomen after eating — shows up in 5–8% of people on tirzepatide. This is notably higher than what you’ll see listed on semaglutide’s prescribing information, and it is one of the key differences between the two molecules’ side-effect profiles. Dyspepsia is more prominent on the tirzepatide label because of how strongly tirzepatide slows stomach emptying through both its GLP-1 and GIP effects. Smaller, more frequent meals, eating slowly, avoiding lying down for at least two hours after eating, and steering clear of carbonated drinks all help. If you also have heartburn (acid rising into your throat), mention it to your doctor — GERD is listed as an adverse reaction in the Zepbound obesity trial data.
Abdominal pain on tirzepatide: what is normal and what is not
Abdominal pain occurred in around 5–10% of people in the tirzepatide clinical programs. Mild cramping or bloating in the first days of a new dose is common and usually resolves. The kind of abdominal pain that warrants same-day attention is different: severe, constant, or intensifying pain — especially if it radiates to the upper back, is accompanied by vomiting, or is associated with yellow skin or eyes. That combination can signal acute pancreatitis or gallbladder disease, both of which are listed as warnings in the prescribing information.
Tirzepatide and hair loss: what the labels actually say
Hair loss differs between the two tirzepatide labels. The Zepbound prescribing information (obesity indication) lists alopecia as an adverse reaction in Table 1 of the SURMOUNT data. The Mounjaro prescribing information (Type 2 diabetes indication) does not list it in Table 1 — it appears only under postmarketing reports (Section 6.2), meaning it was reported by real people after the drug reached the market rather than captured as a statistically significant signal in the controlled T2D trials. Neither label provides a specific percentage.
The biology is the same as with semaglutide or any other method that produces rapid weight loss: significant calorie reduction or metabolic change pushes hair follicles into a resting phase. Three months later, those follicles shed together in a process called telogen effluvium. The hair itself is not permanently damaged — it is on a delayed clock. Regrowth typically starts within three to six months after weight stabilizes and is complete by six to twelve months. The best protective lever is protein: clinical guidelines recommend 1.2 to 1.6 grams per kilogram of body weight daily during active weight loss to keep hair follicles and other lean tissue in a growth phase rather than a starvation mode. A multivitamin with iron and zinc is a reasonable addition if bloodwork shows deficiencies.
Fatigue on tirzepatide: when it peaks and what helps
Fatigue affected 4–7% of people in tirzepatide trials. It tends to cluster in the first 48 hours after each injection and at the time of dose increases, then settles into the background for most people once they are at a stable dose. Two common drivers compound the drug effect: eating significantly less than before (reduced calorie intake means less energy available), and not hitting protein targets (low protein accelerates the lean mass loss that makes fatigue worse). Making sure you eat regularly even on days when appetite is suppressed, and tracking protein specifically, matters more than people expect in the early months.
“I've been on MeAgain for about 3 weeks to help me stay on track with my protein, fiber and water needs while taking my Zepbound. I really like the ease of tracking foods and the fact that I can see my progress.”
Tirzepatide sulfur burps and belching
Belching (including the sulfur-smelling variety people describe online) is listed as an adverse reaction in the Zepbound Table 1 data. The mechanism is straightforward: tirzepatide slows gastric emptying, so partially digested food sits in the stomach longer and produces more gas, including hydrogen sulfide. This is more noticeable at higher doses. Eating smaller meals, avoiding carbonated drinks, not drinking through straws, and eating more slowly all reduce the volume of air and gas entering the digestive system. Most people find this side effect improves significantly once they have been at a stable dose for six to eight weeks.
‘Tirzepatide face’: what causes it and what helps
The term ‘tirzepatide face’ refers to the facial volume loss — hollowed cheeks, more visible temples, and changes under the eyes — that can accompany significant weight loss on this medication. It is not a unique side effect of tirzepatide; it is a class effect of any treatment that produces rapid significant weight change, including the same phenomenon widely called ‘Ozempic face’ with semaglutide. The body reduces fat mass from everywhere, including the fat pads in the face that soften and fill features.
The main protective lever is protein. Hitting the 1.2 to 1.6 g/kg target that clinical weight-loss guidelines recommend keeps more of your weight coming off fat rather than lean tissue, which softens how pronounced the facial change becomes. Slower weight loss — staying at a lower dose longer rather than rushing to the maximum — gives skin more time to adapt. The change is real and sometimes distressing, but it is stable or improvable once weight stabilizes, protein is adequate, and enough time has passed. Dermatologists who work with GLP-1 populations report that most people’s faces normalize within twelve to eighteen months after reaching a steady weight.
Tirzepatide and vision: NAION and diabetic retinopathy risk
Sudden vision change or loss in one eye is a red flag that needs urgent attention regardless of which medication you are on. Nonarteritic anterior ischemic optic neuropathy (NAION) — a blockage of blood flow to the optic nerve — has been confirmed by the European Medicines Agency’s safety review committee as a very rare side effect of semaglutide medicines (Ozempic, Wegovy, Rybelsus). A 2024 retrospective cohort analysis (Hathaway et al., JAMA Ophthalmology 2024) reported an elevated NAION hazard specifically in people prescribed semaglutide compared to matched controls. Tirzepatide has NOT been the subject of an NAION regulatory action; no EMA, FDA, MHRA, or WHO safety signal has linked tirzepatide to NAION. The absolute risk on semaglutide is low, but the consequence of missed NAION is permanent vision loss — which is why any sudden painless vision change in one eye on any GLP-1, especially in the first hours to days after a dose, should prompt a same-day call.
For people with pre-existing Type 2 diabetic retinopathy, the Mounjaro prescribing information (Section 5.8) includes a specific warning: rapid improvement in blood sugar can temporarily worsen retinopathy. This was observed in the SURPASS-1 trial. If you have pre-existing retinopathy and are starting Mounjaro, discuss eye monitoring frequency with your doctor before your first dose.
Hypoglycemia on tirzepatide: low blood sugar and who is at risk
Tirzepatide used alone does not cause meaningful low blood sugar. In SURPASS-1 monotherapy over 40 weeks, blood sugar below 54 mg/dL occurred in 0% of tirzepatide users across all three doses versus 1% on placebo. Tirzepatide stimulates insulin secretion only when blood sugar is already elevated — a glucose-dependent mechanism that essentially turns itself off at normal blood sugar levels. This is why monotherapy hypoglycemia is not a realistic concern. The risk changes substantially when tirzepatide is combined with other diabetes medications.
With basal insulin: blood sugar below 54 mg/dL occurred in 14 to 19% of Mounjaro users depending on dose in SURPASS-5 (Dahl et al., JAMA 2022), versus 13% on placebo plus insulin. With a sulfonylurea (a type of diabetes pill that lowers blood sugar), SURPASS-4 (Del Prato et al., Lancet 2021) reported hypoglycemia below 54 mg/dL in roughly 10 to 14% of tirzepatide users on the combination (9.9% at 10 mg, 12.8% at 15 mg, 13.8% at 5 mg) over up to 104 weeks. These numbers are why your doctor may reduce your insulin or sulfonylurea dose when you start tirzepatide. Signs of low blood sugar include shakiness, sweating, rapid heartbeat, confusion, and feeling faint. Keep a fast-acting carbohydrate accessible and check your blood sugar if you feel off.
Tirzepatide and pancreatitis: what the 2026 warning says
Pancreatitis is listed as a warning in both the Mounjaro and Zepbound prescribing information (Section 5.2). In the SURPASS trial data, acute pancreatitis occurred at approximately 0.23 events per 100 person-years with tirzepatide versus 0.11 with comparators — roughly double the comparator rate in the controlled T2D trials, but small in absolute terms for most people. The picture from real-world post-market safety monitoring has been more serious: on January 29, 2026, the UK MHRA strengthened its pancreatitis warning for all GLP-1 and dual GLP-1/GIP medications to explicitly include necrotizing and fatal cases, based on reported cases since market approval.
The practical warning sign is severe abdominal pain that will not ease — especially if it radiates toward your upper back, is constant rather than crampy, is accompanied by vomiting, or comes with fever. If you experience that combination, go to urgent care or call your doctor the same day. Tirzepatide should be permanently discontinued if pancreatitis is confirmed.
Thyroid concerns: the FDA’s strongest tirzepatide warning
Both the Mounjaro and Zepbound labels carry the FDA’s most serious category of warning about thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). This warning is based on animal studies in rats and mice — tirzepatide produced thyroid C-cell tumors at doses significantly higher than those used in humans, in a dose- and duration-dependent pattern. No direct human cases of MTC caused by tirzepatide have been confirmed, but the FDA and Eli Lilly require the warning because the mechanistic concern applies to the entire GLP-1 class.
Do not take tirzepatide if you or an immediate family member has a personal or family history of medullary thyroid carcinoma or MEN 2 (Multiple Endocrine Neoplasia syndrome type 2). These are hard contraindications, not relative ones. Report any new lump or swelling in the neck, hoarseness, trouble swallowing, or shortness of breath to your doctor promptly — these can be early signs of thyroid changes.
Tirzepatide and gallbladder disease
Gallbladder disease — including gallstones and acute cholecystitis (gallbladder inflammation) — is listed as a warning in the Zepbound prescribing information (Section 5.3). The mechanism is likely related to slowed bile flow from reduced gallbladder contraction, compounded by rapid weight loss (which concentrates bile and promotes stone formation). Yellow skin or eyes, severe right-sided abdominal pain especially after fatty meals, nausea and vomiting accompanying abdominal pain, or fever alongside abdominal discomfort are the warning signs that need same-day evaluation. If you develop confirmed gallstones requiring surgery, tirzepatide will typically need to be stopped while you recover.
Tirzepatide and kidney health: dehydration and acute kidney injury
Acute kidney injury from volume depletion is listed in both the Mounjaro and Zepbound prescribing information (Section 5.5). The mechanism is not a direct toxic effect of tirzepatide on kidneys — it is the dehydration that can result from severe vomiting or diarrhea. In people with Type 2 diabetes or existing kidney involvement, this dehydration-driven kidney injury is a clinically relevant risk. The practical protection is uncomplicated: stay well hydrated, and if GI side effects are severe enough that you cannot keep fluids down for more than 24 hours, contact your doctor the same day.
On the protective side, the SURPASS trial data shows a kidney benefit signal for people with Type 2 diabetes. A post-hoc SURPASS-4 kidney analysis found a meaningful reduction in a composite kidney endpoint compared to insulin glargine. This is one reason tirzepatide is of particular clinical interest in people with T2D and early kidney disease. The FDA label requires no dose adjustment even with severe chronic kidney disease (eGFR as low as 15).
Tirzepatide long-term side effects: what four years of data shows
SURPASS-CVOT (Nicholls et al., NEJM 2025) provides the longest-term safety data for tirzepatide — a cardiovascular outcomes trial with a median follow-up of approximately four years in 13,165 people with Type 2 diabetes and established atherosclerotic cardiovascular disease. In that data, tirzepatide met the prespecified noninferiority bar versus the active comparator (dulaglutide) for major cardiovascular events but did not achieve statistical superiority (event rates were 12.2% vs 13.1%, HR 0.92, 95.3% CI 0.83–1.01, P=0.09), meaning tirzepatide’s cardiovascular safety was at least as good as an established GLP-1 medication. GI side effects remained most common at each dose transition; no new serious long-term signals were identified at the four-year mark.
What is less established: the very long-term (beyond five years) safety of dual GIP and GLP-1 action, and whether the bone density changes seen with significant weight loss on any medication apply to tirzepatide with the same magnitude. For most people, the available four-year SURPASS-CVOT data represents meaningful real-world durability without surprises.
Tirzepatide side effects in women: birth control pill absorption
Women on tirzepatide face one drug interaction that gets less attention than it deserves: the birth control pill. The Mounjaro and Zepbound labels (Section 8.3) note that oral contraceptive absorption may be reduced for four weeks after starting tirzepatide and for four weeks after each dose increase. Tirzepatide slows gastric emptying, which changes how quickly and consistently oral pills are absorbed — the concern is that pill levels in the bloodstream during those transition windows may be lower than expected, reducing contraceptive efficacy.
The FDA label recommends switching to a non-oral contraceptive — an IUD, implant, or patch — OR adding a barrier method during those four-week windows after starting or stepping up the dose. This applies every time the dose increases, not just when you start. If you are on a birth control pill and beginning or increasing tirzepatide, raise this at your next visit before the next injection.
Separately, women with Type 2 diabetes or insulin resistance who have had irregular menstrual cycles may see those cycles normalize or return on tirzepatide, because improved metabolic control can restore ovulation. Fertility can return before a woman is aware her cycle has regularized — another reason reliable contraception is important early in treatment.
Tirzepatide and pregnancy: what to do and when to stop
Based on animal studies, tirzepatide may cause fetal harm. The FDA prescribing information (Section 8.1) for both Mounjaro and Zepbound states that the drug should be discontinued when pregnancy is recognized or planned. If you are considering becoming pregnant, stop tirzepatide at least two months before trying to conceive — the approximately five-day half-life means the drug clears your system fully in about 25 days, and the label’s two-month recommendation provides a safety buffer beyond that. If you discover you are pregnant while on tirzepatide, stop the medication and contact your care team promptly.
There is no established evidence of a direct fertility-enhancing effect from tirzepatide itself. However, improved metabolic health — reduced insulin resistance, restored regular cycles — can make conception more likely in women who were metabolically disrupted before starting. This is worth discussing with your doctor if you are trying to conceive, because resuming medication post-pregnancy is a separate conversation from stopping it before.
“As a girly on a new GLP-1 journey, this app has been such a fun way to track my dosing, symptoms, meals, water, steps and more! The Capy widget is a favorite.”
One practical heads-up that applies to everyone on tirzepatide regardless of why they are taking it: surgery planning is different when you are on a GLP-1.
Tirzepatide and surgery: the anesthesia and aspiration risk
If you have a scheduled procedure requiring general anesthesia or deep sedation, tell your surgical team you are on tirzepatide well before the day of surgery — ideally at the time of scheduling. Tirzepatide significantly slows gastric emptying, which means food and liquid can remain in your stomach even after the standard overnight fast. During anesthesia, this raises the risk of aspiration — stomach contents entering the lungs — a potentially serious or life-threatening complication. The FDA updated both the Mounjaro and Zepbound labels in November 2024 to add pulmonary aspiration during elective procedures as a postmarketing adverse reaction, and the UK MHRA issued a corresponding Drug Safety Update on the same concern.
The UK MHRA issued a formal Drug Safety Update specifically on this concern, and the American Society of Anesthesiologists’ 2024 multi-society guidance states that most people can continue their GLP-1 medication before elective surgery but should follow a liquid-only diet for 24 hours before the procedure. People in the active dose-increase phase or with significant ongoing GI symptoms are at higher risk and may need to pause tirzepatide or delay the procedure. The decision depends on the urgency of the surgery and your individual situation — ask both your prescribing doctor and your surgical team to coordinate before you arrive.
Tirzepatide dose steps and why side effects change at each increase
Both Mounjaro and Zepbound follow the same six-step schedule: 2.5 mg weekly for four weeks (tolerability only), then 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg at minimum four-week intervals. The 2.5 mg starting dose is not a treatment dose — it produces modest appetite suppression but minimal clinical effect. The goal at that level is simply to let your body meet the medication. Each increase resets the adjustment clock: expect days two through five after a step-up to feel louder than the later days of the same dose.
You do not have to move through all six steps on a fixed schedule. Staying at 5 mg or 7.5 mg for an extra month to let GI side effects settle is a well-accepted clinical practice. The four-week minimum between increases is a floor, not a ceiling. If your doctor is monitoring both your outcomes and your tolerability, the decision about when to step up should be a conversation, not an automatic clock.
“I've been on my MJ journey since September 2025. I downloaded this app shortly after I started and it was advertised on my #fyp. What a wonderful decision! Seeing my food intake has made a huge difference in my journey. Also, reviewing my shot locations has helped me to remember to rotate locations. I have recommended this app to my friends who have also started their own journeys to being healthier. Great app!!!”
Injection site reactions on tirzepatide
Injection site reactions — redness, swelling, itching, or a small bruise at the site — occurred in 3.2% of Mounjaro users versus 0.4% of placebo participants in the SURPASS trials, per the prescribing information. In people who developed anti-tirzepatide antibodies, the rate was higher at 4.6%. Reactions are almost always mild and resolve within a few days. Rotating injection sites between the stomach, thigh, and upper arm with each shot prevents localized irritation from building up in one area. If you notice a firm lump that does not resolve within a week, spreading redness that grows over 24 hours, or signs of infection (warmth, discharge, or fever), contact your doctor.
Stopping tirzepatide: what happens to side effects and weight
When tirzepatide is stopped, GI side effects resolve as the drug clears your system over approximately 25 days (five half-lives of roughly five days each). Nausea and appetite suppression typically ease within a week of the last injection. Constipation may linger slightly longer as digestive transit returns to its previous baseline.
Weight regain after stopping tirzepatide is well documented. SURMOUNT-4 (Aronne et al., JAMA 2024) and a 2025 post-hoc analysis showed that cardiometabolic parameters that had improved on tirzepatide — blood sugar, blood pressure, lipids, and weight — largely reversed toward baseline after stopping, in proportion to the weight regained. The SURMOUNT-4 design had people complete 36 weeks on tirzepatide, then randomize to either continue or switch to placebo: those who stopped regained roughly two-thirds of their lost weight over 88 weeks. This is not a failure of the drug; it reflects the ongoing nature of obesity as a chronic condition rather than a one-time treatment. Discussing a long-term plan with your doctor before stopping is important.
Mounjaro vs Zepbound: same molecule, different FDA indications
Mounjaro and Zepbound are the same drug — both are tirzepatide manufactured by Eli Lilly, at the same doses (2.5 mg through 15 mg), with the same six-step escalation schedule, and dispensed in the same KwikPen injector format. The distinction is purely the FDA indication. Mounjaro (approved May 2022) is indicated for blood sugar management in Type 2 diabetes. Zepbound (approved November 2023) is indicated for chronic weight management in adults with obesity or overweight with a weight-related health condition.
The side-effect profiles in the label differ because the underlying clinical trial populations differ. Mounjaro’s label draws primarily on the SURPASS trials (people with Type 2 diabetes, often on background medications). Zepbound’s label draws on the SURMOUNT trials (people with obesity, generally without T2D). Zepbound Table 1 lists alopecia and belching as adverse reactions; Mounjaro Table 1 does not, because those symptoms either were not statistically significant in the T2D population or were lower at the doses used. Insurance coverage often differs substantially: Medicare Part D covers Mounjaro for T2D but, as of 2026, does not generally cover Zepbound for weight management.
Tirzepatide vs semaglutide: side-effect comparison from head-to-head data
SURPASS-2 (Frías et al., NEJM 2021) is the only prospective head-to-head trial comparing tirzepatide directly to semaglutide. It enrolled 1,879 adults with Type 2 diabetes on metformin and compared Mounjaro at 5, 10, or 15 mg against Ozempic at 1 mg (the standard T2D dose). On glycemic control and weight loss, tirzepatide was superior at all three doses. On side effects: GI events were somewhat higher in the tirzepatide arms at the highest doses, consistent with a stronger gastric-emptying effect from the dual GIP and GLP-1 mechanism. Dyspepsia, in particular, is more prominent on the tirzepatide label than on semaglutide’s label.
A non-head-to-head comparison of outcomes (SURMOUNT-1 versus STEP-1): tirzepatide 15 mg produced an average of 20.9% weight loss at 72 weeks in SURMOUNT-1 (n=2,539); semaglutide 2.4 mg produced 14.9% weight loss at 68 weeks in STEP-1 (n=1,961). These are not directly comparable trials (different populations, different designs) but represent the best available public data.
Compounded tirzepatide: 503A vs 503B, regulatory status, and risks
Compounded tirzepatide is prepared by licensed US-based 503A and 503B pharmacies under a valid prescription. It is not FDA-approved as a specific compounded drug product the way Mounjaro and Zepbound are, and the FDA has cautioned that compounded versions are not tested for the same batch-to-batch consistency, sterility, or drug-level predictability as the brand products. There are important regulatory distinctions. Section 503A pharmacies make prescription-specific compounds based on an individual prescription from a licensed clinician. Section 503B outsourcing facilities produce larger batches under stricter FDA oversight. Neither can be described as FDA-approved in the way Mounjaro and Zepbound are — they are compounded preparations, not manufactured drugs, and they fall outside the FDA’s drug approval pathway.
The FDA declared the tirzepatide shortage resolved in 2025 and has since issued warning letters and enforcement actions against compounders and telehealth companies that marketed unauthorized or improperly compounded tirzepatide. At the same time, licensed 503A and 503B pharmacies that operate within the legal framework remain an established and legal pathway to access compounded tirzepatide, particularly for people who cannot afford or cannot access brand-name Mounjaro or Zepbound.
Risks specific to compounded sources: potency variation (compounded preparations are not subject to the same batch consistency testing as manufactured drugs), sterility concerns if quality controls are inadequate, and no systematic post-market safety monitoring. In February 2026, the UK MHRA issued an alert about falsified Mounjaro KwikPen 15 mg pre-filled pens in the UK supply chain. Health Canada issued a counterfeit GLP-1 alert in January 2026. If you use compounded tirzepatide, use only licensed US-based compounding pharmacies that can provide a Certificate of Analysis for each batch and ship within a clinically supervised prescription framework.
MeAgain’s compounded tirzepatide
MeAgain offers compounded tirzepatide at $199 per month with fair pricing through licensed US-based 503A and 503B pharmacies — the same price at every dose from 2.5 mg to 15 mg. This is an optional add-on for eligible subscribers inside the app, not the primary purpose of the app. We make money from the $10/month app subscription. If you want medication access through the app, an in-app intake connects eligible subscribers with a licensed clinician who reviews and manages the prescription.
Gastroparesis and severe GI motility problems on tirzepatide
In December 2025, the FDA updated both the Mounjaro and Zepbound prescribing information to add a specific warning about severe gastroparesis (Section 5.6): tirzepatide is not recommended for people with pre-existing severe gastroparesis. This was added because of reported cases in which severe gastroparesis was exacerbated or precipitated, requiring hospitalization, in people on tirzepatide. Gastroparesis is a condition where the stomach cannot empty itself normally, which is worsened by a drug that deliberately slows gastric emptying further.
The practical implication: if you have a history of gastroparesis or symptoms suggesting poor gastric emptying (persistent nausea, chronic bloating, vomiting of undigested food hours after eating), this needs to be discussed with your doctor before starting tirzepatide or during any dose increase. Ileus — a complete stoppage of bowel movement — is listed as a postmarketing adverse reaction on the Mounjaro label. These are rare but serious, and they reinforce why reporting significant persistent GI symptoms to your doctor matters rather than waiting them out.
Tirzepatide and muscle loss: what body composition data shows
Some muscle loss is expected with any significant caloric deficit and weight loss, including on tirzepatide. In the SURMOUNT-1 body composition substudy at 15 mg over 72 weeks, fat mass decreased by approximately 33.9% from baseline and lean mass decreased by approximately 10.9% from baseline — which works out to roughly three-quarters of total weight lost coming from fat and the remainder from lean tissue and fluid. This ratio is broadly consistent with other weight-loss methods — not a unique fault of tirzepatide, but a reminder that the weight that comes off is not all fat.
The two evidence-backed levers to protect lean mass are protein intake and resistance training. Clinical guidelines recommend 1.2 to 1.6 grams of protein per kilogram of body weight per day during active weight loss. Without those inputs, a larger share of weight lost comes from muscle rather than fat, which worsens fatigue, slows metabolism, and affects long-term body composition. As MeAgain reviewer caressita put it: “it showed me I should have more protein and water intake and it logs all my foods.” MeAgain tracks protein alongside your shot schedule specifically because this matters — and because tirzepatide’s appetite suppression makes it easy to undereat protein even when overall food intake is already low.
“I was a little hesitant getting an app at first I've talked to a few other people who have done GLP and didn't use an app but I personally found it super helpful, especially with my shot tracking as sometimes I can forget where I had the shot the week before so it's nice to keep a track record and rotate correctly! I also love the little widget buddy that shows your tracking of water, exercise, food, fiber it's and easy way to see it and be like oh you're right I should have a healthy snack especially when you're in the first few days of your shot and aren't hungry and can easily forget to eat. I've been on it for 120 days and it's been great!”
Tirzepatide and alcohol: how tolerance changes
Alcohol is not contraindicated on tirzepatide, but most people find their tolerance shifts significantly on the medication. Tirzepatide slows gastric emptying, so alcohol moves from the stomach into the bloodstream more slowly but then hits all at once once that window opens — producing stronger effects, often with less warning than before. The delayed and then more intense alcohol response catches many people off guard in the first few months on the medication. Nausea and its aftermath are also more likely for the 24 to 48 hours after drinking.
For people with Type 2 diabetes on insulin or a sulfonylurea, alcohol adds an additional concern: it can mask hypoglycemia symptoms and independently lower blood sugar, creating a more unpredictable risk than either alone. Practical approach: eat a protein-containing meal before drinking, stick to lower-alcohol options in smaller volumes, avoid sugary cocktails, hydrate between drinks, and check blood sugar if you are on diabetes medication. Most people find their natural drinking volume drops on tirzepatide — appetite suppression extends to the appetite for alcohol for many people.