How long do Mounjaro side effects last?
For most people on Mounjaro, GI side effects are worst in the first month and ease noticeably by weeks six to eight. Nausea typically peaks in the two to four days after each dose increase, then softens as tirzepatide blood levels stabilize. Because tirzepatide takes about five days to drop by half, your body needs roughly two to three weeks at each new dose before levels fully settle — which explains why the FDA label builds in a gradual six-step schedule from 2.5 mg to a 15 mg maximum, with increases no faster than every four weeks. The 2.5 mg starting dose is a tolerability dose, not a treatment dose. At that level, many people feel modest appetite suppression but minimal GI disruption.
In the SURPASS-1 trial (Rosenstock et al., Lancet 2021), people on tirzepatide in a controlled Type 2 diabetes population saw GI event rates cluster in the dose-increase months, with rates declining for participants who stayed on the drug past the initial adjustment period. The clinical takeaway: if you’re in week one or two and wondering whether to push through, what you’re feeling is expected and time-limited. Stopping before the adjustment curve turns is where most discontinuations happen.
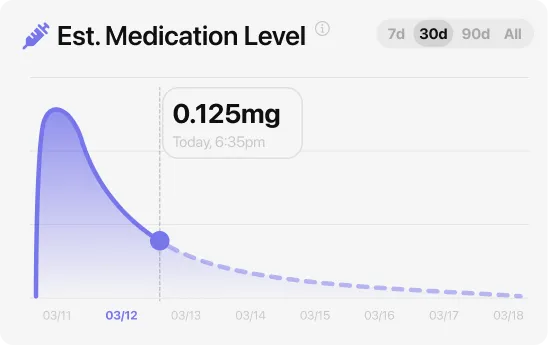
One pattern that comes up in MeAgain reviews: people who log their symptoms alongside what they ate and when they injected find the adjustment period easier to navigate. The data makes the pattern visible instead of something you’re guessing at week by week.
Which Mounjaro side effects are serious?
Most Mounjaro side effects are uncomfortable but not dangerous. A smaller group are genuine red flags that need same-day attention: acute pancreatitis, gallbladder disease (including gallstones), severe allergic reactions, and kidney injury from dehydration. The FDA Mounjaro prescribing information (Section 5.1) carries the agency’s strongest warning about thyroid C-cell tumors seen in rat studies — do not take Mounjaro if you or a family member have a history of medullary thyroid cancer or MEN 2 (a rare inherited hormone condition). A postmarketing update from December 2025 added severe gastroparesis as a new warning (Section 5.6): Mounjaro is not recommended for people with severe gastroparesis.
The most important warning sign is severe, persistent stomach pain — especially if it bores through to your upper back, or if you also have vomiting that won’t stop or yellowing of the skin or eyes. These can signal pancreatitis or gallstones, both of which require a doctor that day, not next week. In the SURPASS trials, acute pancreatitis occurred at 0.23 per 100 person-years with tirzepatide versus 0.11 with comparators — small in absolute terms, but roughly double the comparator rate, which is why the FDA label calls it out specifically.
For people with Type 2 diabetes, volume depletion from GI side effects — vomiting or diarrhea leading to dehydration — can trigger acute kidney injury (Section 5.5 of the label). This is a specific concern in the T2D population because many people with T2D are already on medications that can stress the kidneys. Staying hydrated is not just comfort advice; it is a clinical precaution.
!Call your doctor right away if you notice any of these
- Severe stomach pain that won’t go away
- pain that moves through to your upper back
- vomiting that lasts more than 24 hours
- yellow skin or eyes
- shakiness, sweating, or confusion (especially if you’re also on insulin)
- a lump or swelling in your neck
- trouble breathing or swallowing
- a rash with swelling of the face, lips, or tongue
- persistent nausea or vomiting at a stable dose that has not improved over several weeks.
Hypoglycemia on Mounjaro: low blood sugar and what to watch for
Mounjaro used alone rarely causes low blood sugar. In the SURPASS-1 monotherapy trial (40 weeks), the rate of blood sugar below 54 mg/dL was 0% across all tirzepatide doses versus 1% for placebo — meaning Mounjaro on its own is not a meaningful hypoglycemia driver. That picture changes when Mounjaro is combined with other diabetes medications.
With basal insulin (40 weeks): blood sugar below 54 mg/dL occurred in 16% of people on 5 mg, 19% on 10 mg, and 14% on 15 mg, compared to 13% on placebo plus insulin. With a sulfonylurea (a type of diabetes pill that lowers blood sugar) over 104 weeks: rates were 13.8% at 5 mg, 9.9% at 10 mg, and 12.8% at 15 mg. These are not trivial numbers, and they are exactly why your endocrinologist may reduce your insulin or sulfonylurea dose when Mounjaro is added to your regimen.
Signs of low blood sugar include shakiness, sweating, fast heartbeat, hunger, confusion, or feeling faint. Keep a fast-acting carbohydrate accessible — glucose tablets, juice, or regular soda — and test your blood sugar if you feel off. If someone with you seems confused or loses consciousness, that is a medical emergency: call 911. If you are using Mounjaro primarily for weight management without Type 2 diabetes, the risk of low blood sugar from tirzepatide alone is very low.
What’s the best way to manage Mounjaro nausea?
Three tactics that work for most people: eat small protein-first meals on shot day, hydrate consistently, and slow the pace of dose increases if your care team agrees. Nausea on Mounjaro is strongest when your stomach is empty or when you eat large, high-fat meals — so the move is to eat before you feel hungry, keep portions small, and lead with protein at every meal.
The protein-first strategy
- Eat a small protein-led meal one to two hours before your shot. Greek yogurt, eggs, a protein shake, or chicken and rice all work.
- Sip water throughout the day. Aim for at least 64 oz; more on shot days or when you’re dealing with diarrhea.
- Avoid fried food, very greasy meals, and large portions for 48 hours after each shot — tirzepatide slows stomach emptying, so heavy food amplifies nausea.
- Try ginger (tea, candies, or capsules) or peppermint for mild queasiness. Both have evidence for reducing nausea and carry low risk.
- If nausea is still strong at week three, ask your doctor whether holding at your current dose for an extra month makes sense before stepping up.
Hydration targets on Mounjaro
Sixty-four ounces is a floor, not a ceiling. On shot days and for the 48 hours after, many people on Mounjaro find they need closer to 80 to 90 ounces to keep nausea and headaches in check. In people with Type 2 diabetes, adequate hydration also reduces the risk of kidney strain from dehydration that the FDA label specifically flags. A practical routine: fill a 32-ounce bottle in the morning, refill at lunch, refill again in the afternoon. If your urine is dark yellow, you’re behind. Electrolyte tablets can help if plain water feels difficult to tolerate during the roughest days.
“This app has helped me while on Trizeptide it showed me I should have more protein and water intake and it logs all my foods I have lost about of weight using this app with medication”
Hydration and nutrition are the two tolerability levers you actually control day to day. The next three H2 sections cover Section 5 safety items on the FDA Mounjaro label: diabetic retinopathy, pancreatitis, and kidney injury from dehydration. These are rarer than the GI side effects covered above but have red-flag patterns to recognize.
Can Mounjaro affect your vision? Diabetic eye disease explained
Yes, and this is one side effect that is genuinely specific to the Type 2 diabetes population rather than a general GLP-1 concern. The FDA Mounjaro label (Section 5.7) includes a warning about worsening diabetic retinopathy in people who already have it before starting Mounjaro. If you have any history of diabetic eye disease, your doctor should monitor for progression after you begin.
The counterintuitive mechanism: rapid blood sugar improvement — which is exactly what Mounjaro is supposed to produce — can paradoxically worsen pre-existing retinopathy in the short term. The eye’s blood vessels, which have adapted to higher glucose levels over time, struggle with the sudden change. In SURPASS-4, which enrolled people with Type 2 diabetes and high cardiovascular risk, retinopathy events were monitored carefully. This is not a reason to avoid Mounjaro if your endocrinologist recommends it, but it is a reason to get a baseline eye exam before starting and follow up every three to six months if you have any degree of pre-existing diabetic eye disease.
A separate, rarer concern is sudden loss of blood flow to the optic nerve, which can cause vision loss in one eye (doctors call this NAION). The European Medicines Agency’s safety review committee concluded in June 2025 that NAION is a very rare side effect of semaglutide medicines (Ozempic, Wegovy, Rybelsus) specifically. Tirzepatide has not been the subject of an NAION regulatory action — no FDA, EMA, MHRA, or WHO safety signal has linked tirzepatide to NAION. That said, sudden vision change on any GLP-1 is an emergency regardless of which drug you are on: any blurring in one eye, loss of part of your visual field, or sudden vision loss needs same-day care. Do not wait to see if it resolves.
Does Mounjaro cause diarrhea?
Mounjaro diarrhea affects 12 to 17% of people per the FDA prescribing information — higher than the 8.5 to 8.8% rate seen with semaglutide (Ozempic/Wegovy) in the SUSTAIN trials. The higher rate in tirzepatide is partly attributed to its dual GIP and GLP-1 action, which slows the stomach more than drugs that only hit GLP-1. Diarrhea usually appears in the first one to five days after starting or stepping up, and is almost always mild to moderate. The most common triggers are high-fat meals and sugar alcohols — sorbitol, xylitol, and erythritol are common in protein bars and sugar-free snacks, so check labels. Stay hydrated, cut back on fatty food for 48 hours after each shot, and eat bland foods — rice, bananas, toast, boiled potatoes — until it passes.
In people with Type 2 diabetes, diarrhea carries a specific secondary concern: dehydration compounds the risk of kidney stress that the FDA label already flags. If diarrhea is bloody, lasts more than three days, or comes with dehydration symptoms (dark urine, dizziness, dry mouth), call your doctor that day. Do not wait and hope it resolves on its own.
Does Mounjaro cause vomiting?
Mounjaro vomiting affects 5 to 9% of users per the FDA prescribing information Table 1, with slightly higher rates at the 10 mg dose and above. Most vomiting episodes happen in the first three to five days after a new dose and almost always resolve within a week. The mechanism is the same as nausea — tirzepatide slows gastric emptying, so food sits longer than your body expects, and when the stomach volume hits a threshold, the body expels it. Vomiting after Mounjaro is not usually a sign that something is wrong; it is your stomach reacting to the slower transit time.
If you vomit once or twice after a dose and feel better, no action is needed beyond rehydrating. If vomiting persists beyond 24 hours, if you cannot keep liquids down, or if it comes with severe abdominal pain, call your doctor that day — these can signal dehydration, pancreatitis, or gallstones. To reduce recurrence, eat smaller bland meals for the first 48 hours after each shot, avoid high-fat food, and take your dose at night so nausea and vomiting peak while you sleep. Persistent vomiting at a stable dose is worth a conversation with your care team about slowing the next dose step.
Does Mounjaro cause sulfur burps?
Yes — sulfur burps are one of the most widely reported real-world side effects of Mounjaro, even though they do not appear as a named item in the FDA label’s adverse-reaction table. No formal incidence study exists; user self-reports in community surveys and FAERS voluntary post-marketing reports consistently describe the symptom, most often during dose increases. The symptom is absent from the FDA Table 1 list because it is not individually coded in the SURPASS trial case report forms, not because it is rare. The mechanism is straightforward: tirzepatide slows gastric emptying significantly, so food sits in the stomach longer. Bacteria ferment proteins that haven’t yet moved through, producing hydrogen sulfide gas, which causes the distinctive egg-like smell.
The worst triggers are high-protein foods (especially eggs), high-fat meals, carbonated drinks, and high-sulfur vegetables like broccoli, cauliflower, and cabbage. Sulfur burps tend to be worst in the first two to four weeks at each new dose and improve as your body adjusts to the slower transit time. They are not dangerous, but they can be socially disruptive. Practical management: eat smaller meals spaced more evenly through the day, avoid carbonated beverages, temporarily cut back on high-sulfur foods during the adjustment window, and stay well hydrated to move food through more consistently. Most people find the problem fades substantially once their body settles at a stable dose.
Does Mounjaro cause constipation?
Mounjaro constipation affects 6 to 7% of users per the FDA prescribing information. Tirzepatide slows gastric emptying — the same mechanism that keeps you full longer — which also slows transit through the colon and can dry out stool. The postmarketing safety update in December 2025 (Section 6.2) added severe constipation, including fecal impaction, as a reported event. This does not mean severe constipation is common, but it means routine constipation should be managed actively rather than ignored.
The fix is straightforward: more water, more fiber (25 to 35 grams a day from vegetables, legumes, chia seeds, or ground flax), and daily walking. A practical approach: add one high-fiber food per meal every few days rather than jumping from a low intake to 30 grams overnight, which usually causes bloating that compounds the discomfort. If fiber and water alone are not enough, ask your pharmacist about an over-the-counter stool softener or osmotic laxative. If you go more than three days without a bowel movement or develop severe abdominal pain, call your doctor.
Does Mounjaro cause dyspepsia (indigestion)?
Dyspepsia — a persistent or recurring sensation of stomach discomfort, bloating, or early fullness during a meal — affects 5 to 8% of people at the lower Mounjaro doses per the FDA prescribing information Table 1. This is notable because dyspepsia appears prominently in Mounjaro’s clinical trial data but is not a top-listed reaction in semaglutide’s label in the same way. The difference likely reflects Mounjaro’s dual action on both GIP and GLP-1 receptors, which affects how the stomach stretches and empties differently than drugs that only target GLP-1.
Practical management is similar to nausea: smaller meals, protein first, avoiding high-fat foods and carbonated beverages, and eating more slowly. If dyspepsia is severe or persistent — lasting beyond four weeks at a stable dose or interfering with your ability to eat — mention it at your next check-in. Your care team may slow the escalation pace or suggest an over-the-counter antacid for relief. Severe or worsening indigestion that does not respond to these measures warrants a call rather than waiting for the next scheduled appointment.
Does Mounjaro cause hair loss?
Hair shedding on Mounjaro appears in the FDA prescribing information under postmarketing adverse reactions (Section 6.2), meaning it was reported by real people after the drug reached market rather than inside the controlled SURPASS trials. The label does not give a percentage. It is real, but the exact rate in a general population is harder to pin down than the GI side effects.
The mechanism is stress-triggered hair shedding (called telogen effluvium in dermatology). Any rapid, sustained weight loss — surgery, a crash diet, or a GLP-1 medication — pushes a larger share of hair follicles into the resting phase at once, and about three months later those hairs fall out together. It looks dramatic but is almost always reversible. Once weight stabilizes, follicles return to normal cycling, typically within six to twelve months.
Two things consistently reduce severity: hitting your daily protein target and avoiding very-low-calorie days. Clinical guidelines recommend 1.2 to 1.6 grams of protein per kilogram of body weight per day during active weight loss. When the body is running short on both calories and protein, it prioritizes vital organs over hair follicles. A multivitamin with iron and zinc is reasonable if bloodwork shows low levels, since both minerals support the hair growth cycle.
Does Mounjaro cause fatigue?
Fatigue is listed as a common adverse reaction in the FDA Mounjaro prescribing information. The SURPASS trial program reported fatigue in roughly 4 to 6% of participants, with slightly higher rates at the 10 and 15 mg doses. It is usually most noticeable in the first two to four weeks of a new dose and improves as your body adjusts. The tiredness is often indistinguishable from what you would feel eating significantly less food than usual — because that is a large part of what is happening.
The three biggest drivers of Mounjaro fatigue are dehydration, under-eating, and low blood sugar (if you are also on insulin or a sulfonylurea). Hitting your daily protein target, drinking enough water, and eating small meals throughout the day instead of skipping them are the three interventions that consistently help. If fatigue is severe, interferes with work, or persists beyond four weeks at a stable dose, ask your doctor for a blood panel — GLP-1 medications can mask iron or B12 deficiency by reducing appetite for the foods that carry those nutrients.
What is ‘Mounjaro face’ and does the FDA list it?
‘Mounjaro face’ is not in the FDA label. It is a colloquial term for the hollowed or gaunt appearance some people notice in their face during significant weight loss. It is not unique to Mounjaro or any GLP-1 medication. Rapid loss of body fat reduces facial volume everywhere, including the cheeks, temples, and under-eye area, where the fat pads that soften the face gradually disappear. The same effect is widely called ‘Ozempic face’ in the media, but the mechanism is identical — any method that produces meaningful weight loss can produce it.
The main protective lever is protein intake. When the body is in a calorie deficit without enough protein, it pulls from lean tissue, which accelerates the appearance of facial wasting. Hitting your daily protein target consistently matters more than the exact timing. Slower weight loss — staying at a lower dose longer rather than rushing to the maximum — also gives skin more time to adapt. The change is reversible to a large degree once weight stabilizes and protein intake is adequate, though some people seek cosmetic dermatology to speed recovery of facial volume.
Are Mounjaro side effects different in women?
Mounjaro side-effect rates do not differ meaningfully between men and women in the SURPASS trial data. FDA Table 1 prevalence numbers — nausea 12 to 18%, diarrhea 12 to 17%, vomiting 5 to 9%, constipation 6 to 7% — are reported across the full trial population and were not stratified by sex in the primary analyses. Real-world user reports suggest women may experience slightly more nausea and hair shedding, but the difference is small and has not been established in controlled data.
Two considerations matter more for women on Mounjaro than for men. First, the FDA label recommends switching from oral contraceptives to a non-oral method (implant, IUD, or patch) or adding a barrier method for the four weeks after starting Mounjaro and after each dose increase, because slower gastric emptying can reduce absorption of the pill. Second, Mounjaro is not recommended during pregnancy and should be stopped at least two months before trying to conceive. Ovulation can return quickly as weight decreases in people who had weight-related anovulation, so effective contraception matters even if you were not previously fertile.
What are the long-term side effects of Mounjaro?
Long-term Mounjaro safety data comes from the SURPASS trial program, which followed participants for up to 104 weeks, and SURPASS-CVOT (Nicholls et al., NEJM 2025), which followed 13,165 adults for a median of 4 years. No new safety signals emerged in that period beyond what the shorter trials had already shown. The most common side effects — GI symptoms — continue to decline in frequency and severity after the first few months as the body adjusts to slower gastric transit.
The longer-term concerns worth watching are the ones the FDA label flags in its Warnings section: thyroid C-cell tumors (seen in rat studies, not confirmed in humans), pancreatitis (small absolute increase over comparators), gallbladder disease, and worsening diabetic retinopathy in people with pre-existing retinopathy. None of these are guaranteed; most are rare. The practical long-term plan is the same as the first-year plan: stay at the lowest effective dose, keep up annual bloodwork including A1C and thyroid, get an eye exam yearly if you have diabetes, and tell any new doctor you start seeing that you are on Mounjaro.
Is Mounjaro for weight loss or diabetes? Understanding the Zepbound/Mounjaro split
Mounjaro and Zepbound are the same drug — both are tirzepatide, made by Eli Lilly, at identical doses (2.5 mg through 15 mg). The difference is the FDA indication. Mounjaro is approved for Type 2 diabetes management, approved in 2022. Zepbound is approved for chronic weight management in adults with obesity or overweight plus a weight-related condition, approved in 2023. Both use the same injector pen, the same six-step dose schedule, and the same clinical pharmacology.
This distinction matters practically in several ways. First, insurance: Medicare Part D covers Mounjaro for Type 2 diabetes but does not cover Zepbound for weight management (as of the time of publication — coverage rules can change). Commercial insurance coverage depends on the plan. Second, the clinical trial evidence: Mounjaro’s approval is backed by the SURPASS trials in T2D populations. Zepbound’s approval is backed by the SURMOUNT trials in obesity populations. The side-effect profile is the same drug in both cases, but the diabetes-specific risks (hypoglycemia, retinopathy, kidney effects) are more prominent in the Mounjaro/T2D literature.
If you are using Mounjaro off-label for weight loss without Type 2 diabetes, the prescribing clinician may be choosing it because Zepbound coverage or access is unavailable, or because the dose schedule is identical and there is no clinical difference. That is common in practice. Always follow your prescribing doctor’s guidance on indication and insurance eligibility — the decision to use Mounjaro off-label belongs with your clinician, not a tracking app. Just be aware that the side-effect data most directly applicable to you comes from the SURMOUNT trial program — specifically the 20.9% average weight loss at 15 mg in SURMOUNT-1 (Jastreboff et al., NEJM 2022) — rather than the SURPASS diabetes trials.
How do Mounjaro side effects compare to Ozempic? The SURPASS-2 head-to-head data
SURPASS-2 (Frías et al., NEJM 2021) is the only head-to-head trial directly comparing tirzepatide to semaglutide in the same population. It enrolled 1,879 adults with Type 2 diabetes inadequately controlled on metformin and randomized them to Mounjaro at 5, 10, or 15 mg versus Ozempic at 1 mg — the standard diabetes dose, not the 2.4 mg Wegovy weight-loss dose.
On blood sugar (A1C) reduction: Mounjaro outperformed semaglutide at every dose. A1C dropped by 2.01% at 5 mg, 2.24% at 10 mg, and 2.30% at 15 mg, versus 1.86% with semaglutide 1 mg. All three differences were statistically significant (P < 0.001) and met the pre-specified bar for superiority. On weight: Mounjaro produced 1.9 kg more weight loss at 5 mg, 3.6 kg more at 10 mg, and 5.5 kg more at 15 mg compared to semaglutide.
On GI side effects, the two drugs have a similar profile — nausea, diarrhea, vomiting, and decreased appetite — but Mounjaro’s rates are somewhat higher, consistent with its stronger effect on gastric emptying. Mounjaro also carries a dyspepsia signal (5 to 8%) not prominently listed in the semaglutide label, consistent with tirzepatide’s additional GIP receptor action. Hypoglycemia rates in SURPASS-2 were low across all arms (no added insulin or sulfonylurea), which reflects the glucose-dependent mechanism both drugs share.
“I am back with an update since my last review. Patches and updates within the last few weeks have corrected the issues I was having on Thursdays specifically. This app has been a true blessing since the fixes. I take shots for allergies that I also have to alternate and trying to remember where my previous shot was the past few weeks is impossible with 2 different medications. Where ever I take my glp-1 I do an opposite for my allergy medication so it helps me track both. One thing I’d change, I wish I could omit the days I didn’t take photos so that my slideshow would just be the days I took pictures without blank spaces in between.”
What do the first two weeks on Mounjaro actually feel like?
The first two weeks are the adjustment window. Most people notice reduced appetite and mild nausea within 24 to 48 hours of the first 2.5 mg shot, with the roughest days usually falling between days two and five. By the end of week two, tirzepatide levels have stabilized enough that nausea tends to soften from a wave into a background hum.
The 2.5 mg starting dose is not a therapeutic dose — it is a tolerability dose, meant to introduce your body to tirzepatide gradually rather than produce substantial blood sugar or weight change. Some people feel nothing at 2.5 mg and wonder if the shot worked. Others feel significant appetite suppression or mild nausea right away. Both are normal responses to the same starting dose.
Days two through five are typically the hardest in the first month. Tirzepatide levels are climbing toward their weekly peak, appetite is more suppressed than you expected, and food that used to be appealing can suddenly feel unappealing or nauseating. The protein-first strategy matters most here: something small every three to four hours — Greek yogurt, eggs, a protein shake, cheese and crackers — so your stomach is never completely empty.
“I was a little hesitant getting an app at first I’ve talked to a few other people who have done GLP and didn’t use an app but I personally found it super helpful, especially with my shot tracking as sometimes I can forget where I had the shot the week before so it’s nice to keep a track record and rotate correctly! I also love the little widget buddy that shows your tracking of water, exercise, food, fiber it’s and easy way to see it and be like oh you’re right I should have a healthy snack especially when you’re in the first few days of your shot and aren’t hungry and can easily forget to eat. I’ve been on it for 120 days and it’s been great!”
How Mounjaro dose steps affect side effects (2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg)
Side effects tend to be strongest right after each dose increase and get progressively milder as your body adjusts. The pattern typically repeats with each increase but with diminishing intensity — the body adapts progressively.
| Dose step | Duration | What to expect |
|---|---|---|
| 2.5 mg | 4 weeks (tolerability dose) | First nausea and appetite change. Minimal blood sugar or weight impact — this dose builds tolerance, not treatment effect. |
| 5 mg | 4+ weeks | First treatment dose. Blood sugar improvement typically begins. Nausea often peaks in the first one to two weeks after stepping up. |
| 7.5 mg | 4+ weeks | Appetite suppression strengthens. Some people find GI symptoms ease here after adjusting to 5 mg; others see a brief flare. |
| 10 mg | 4+ weeks | Strong appetite suppression. Weight loss accelerates for most people. Hair shedding may begin around this step. |
| 12.5 mg | 4+ weeks | Intermediate step to maximum dose. GI side effects often fade substantially here for people who tolerated prior steps well. |
| 15 mg | Maintenance (if needed) | Maximum approved dose. Used when lower doses aren’t achieving blood sugar or weight goals. In SURPASS-1, 15 mg produced the strongest A1C and weight results. |
Source: FDA Mounjaro prescribing information (2025), Dosage and Administration section. Increase dose in 2.5 mg increments no sooner than every 4 weeks. In SURPASS-1, the 15 mg dose produced A1C reduction of −2.07% and weight loss of −9.5 kg over 40 weeks in people with Type 2 diabetes.
Mounjaro and surgery: anesthesia, aspiration, and what your surgical team needs to know
If you have a scheduled surgery or procedure requiring general anesthesia, tell your surgical team you are on Mounjaro well before the day of the procedure. Because tirzepatide significantly slows gastric emptying, food or liquid can remain in your stomach even after the standard overnight fast, raising the risk of aspiration — stomach contents entering the lungs — during anesthesia. The FDA Mounjaro prescribing information added pulmonary aspiration during elective procedures as a postmarketing adverse reaction in December 2025. The UK MHRA issued a formal safety update on this concern in January 2025.
Updated 2024 multi-society guidance from the American Society of Anesthesiologists and several other medical organizations says that most people can continue their GLP-1 medication before elective surgery, but should follow a liquid-only diet for 24 hours before the procedure. People in the active dose-increase phase or with ongoing significant GI symptoms are at higher risk and may need to pause Mounjaro or delay the procedure — the decision depends on the urgency of the surgery and your individual clinical situation. Ask both your prescribing doctor and your surgical team what the right call is for you specifically. This is not a reason to skip a needed procedure; it is a reason to communicate clearly and early.
Mounjaro and pregnancy: fertility, birth control, and what to know
Based on animal studies, Mounjaro may cause fetal harm. The FDA Mounjaro prescribing information (Section 8.1) states that the drug should be discontinued when pregnancy is recognized. Women who could become pregnant should use reliable contraception while on Mounjaro. This is also relevant because Mounjaro can restore ovulation in women with Type 2 diabetes or insulin resistance whose cycles were irregular — fertility can return before a woman realizes her cycle has normalized.
A critical practical detail (Section 8.3): the birth control pill may be less reliable for four weeks after starting Mounjaro and four weeks after each dose increase. Tirzepatide slows gastric emptying, which changes how quickly and consistently oral pills are absorbed. The FDA label recommends switching to a non-oral method — an IUD, implant, or patch — OR adding a barrier method for those windows. If you use a birth control pill and are starting or stepping up Mounjaro, raise this at your next check-in before the next injection.
If you are planning a pregnancy, the guidance is to stop Mounjaro at least one month before trying to conceive, based on the drug’s approximately five-day half-life and its clearance profile over roughly 25 days. If you discover you are pregnant while on Mounjaro, stop the medication and contact your care team promptly.
Mounjaro and your kidneys: risk and protection
Mounjaro’s relationship with the kidneys is a two-sided story. On the risk side: GI side effects (vomiting, diarrhea) can cause significant fluid loss. In people with Type 2 diabetes, this volume depletion can worsen kidney function, and the FDA label specifically lists acute or worsening kidney failure as a postmarketing adverse reaction (Section 6.2). The practical protection is straightforward: stay well hydrated, and if vomiting or diarrhea is severe enough that you can’t keep fluids down, contact your doctor.
On the protection side, a post-hoc kidney analysis of SURPASS-4 data (Heerspink et al., Lancet Diabetes & Endocrinology 2022) found a striking kidney benefit in people with Type 2 diabetes and high cardiovascular risk. Over the 104-week trial, Mounjaro reduced a composite kidney endpoint by 42% compared to insulin glargine (HR 0.58). A separate pooled analysis across SURPASS-1 through SURPASS-5 found reductions in urinary albumin-to-creatinine ratio (a marker of kidney damage) ranging from roughly 19% to 26% across the three doses compared to pooled comparators. Crucially, the FDA label requires no dose adjustment even in people with severe chronic kidney disease (eGFR as low as 15). This kidney-protective signal is one of the clinically meaningful arguments for Mounjaro specifically in people with T2D and existing kidney involvement.
How to talk to your doctor about Mounjaro side effects
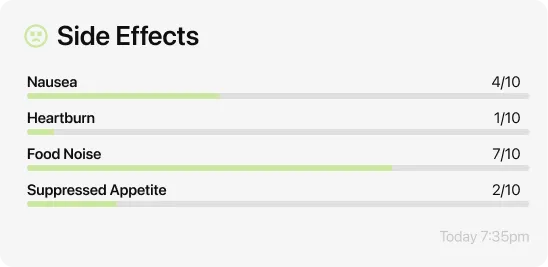
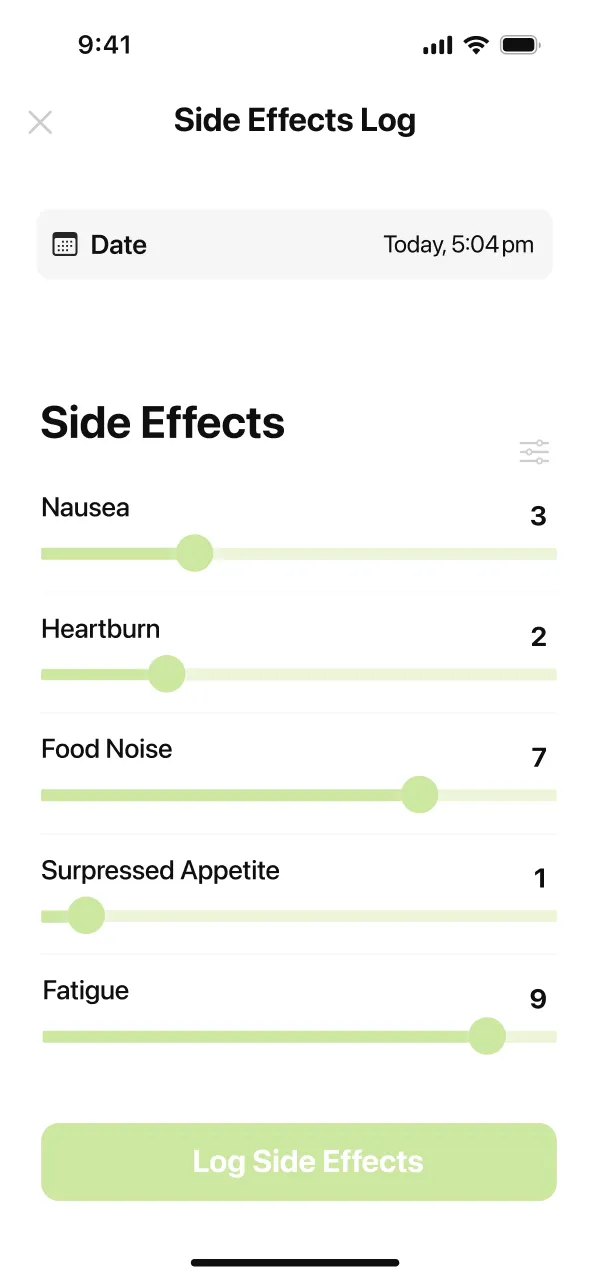
Three things your doctor actually wants to know: which symptoms you’re experiencing and how severe they are on a scale of one to ten, whether they are improving or worsening compared to last week, and whether any of them are affecting your daily life enough that you’re considering stopping.
If you have Type 2 diabetes, your doctor also wants your blood sugar log alongside the side-effect report. Combining GI side effects with lower food intake can shift blood sugar in unexpected directions, and having the data in front of both of you makes dose decisions faster and safer.
Useful phrases that cut through the appointment: “Nausea is running about a five this week, which is better than last week’s seven.” Or: “I’m having trouble eating enough protein because of the nausea — should we hold at this dose longer?” Or: “My blood sugar has been running lower than usual on shot day — should we adjust my insulin dose?” Direct, specific, and tied to a question a care team member can actually answer.
When your symptoms, meals, and shot timing are already logged, you walk into that appointment with a summary instead of trying to reconstruct the past two weeks from memory. That’s the difference between a useful visit and one where you leave without clear answers.
“Love this APP!! Keeps track of ur journey. Very easy to use. I love that if u forget where u did ur shot this keeps track so u know. Tracking my food intake is great too because if I get any medication side effects I can just check here to see what I ate & if that exasperated the symptoms. Everyone on GLP1 should use that APP!!”
What about Mounjaro injection site reactions?
Injection site reactions — redness, swelling, itching, or a small bruise at the injection site — occurred in 3.2% of Mounjaro users versus 0.4% of placebo participants in the SURPASS trials, per the FDA prescribing information. In people who developed anti-tirzepatide antibodies, the rate was higher at 4.6%. Reactions are almost always mild and clear within a few days. Rotating between your stomach, thigh, and upper arm helps prevent localized irritation from building up in one spot. If you notice a firm lump that doesn’t resolve, spreading redness that grows over 24 hours, or signs of infection (warmth, discharge, fever), contact your doctor.
What happens to side effects if you stop Mounjaro?
If you stop Mounjaro, GI side effects resolve as tirzepatide clears your system — which takes about 25 days to clear fully given the approximately five-day half-life (tirzepatide stays in your system for about five days before dropping by half). Nausea and appetite suppression typically ease within a week of the last injection. Constipation may linger slightly longer as digestive transit returns to its previous pace.
For people with Type 2 diabetes, the more pressing concern with stopping is blood sugar rebound. Mounjaro provides meaningful glycemic control, and stopping without an adjusted plan for your other diabetes medications can cause A1C to climb again. If you are considering stopping Mounjaro, talk to your doctor about a transition plan rather than stopping abruptly. Weight regain is also a well-documented pattern after stopping GLP-1 medications from any reason, as appetite and food intake return to previous levels. The blood sugar and weight management effects are medication-dependent, not permanent.