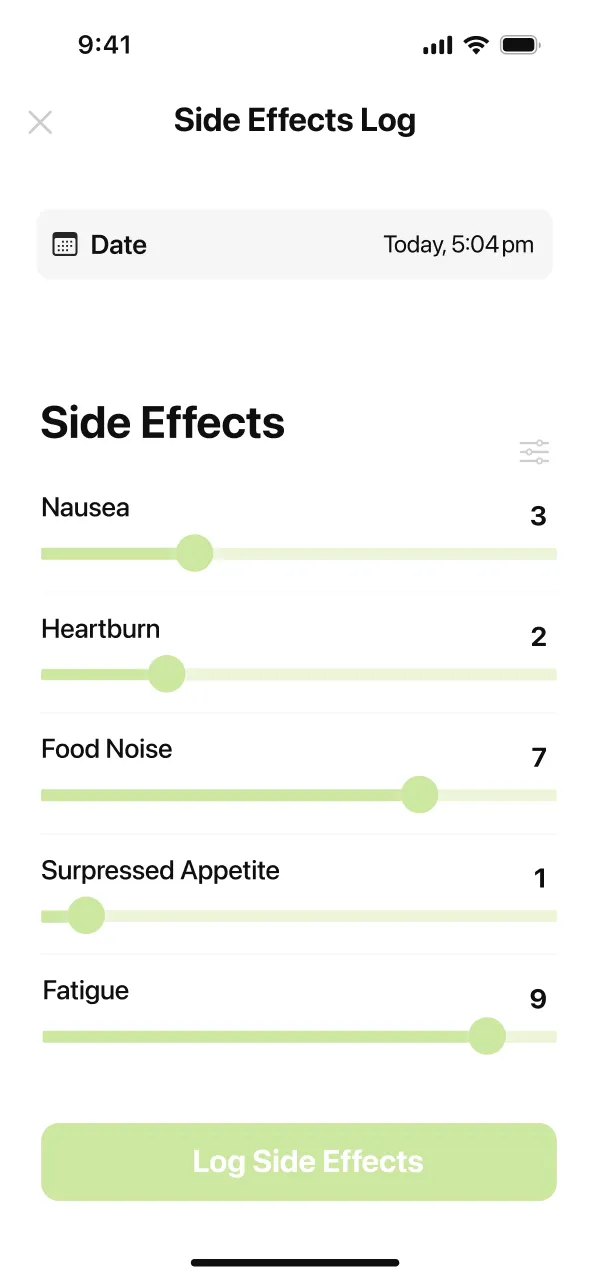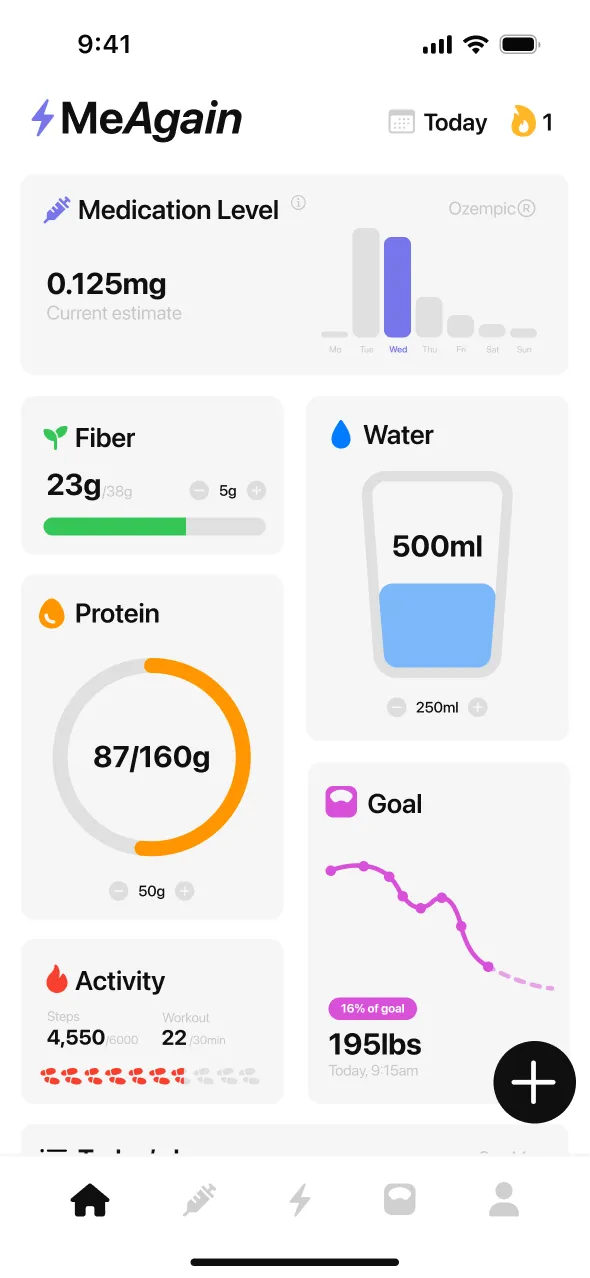
Where does retatrutide stand right now? (Trial-only, not FDA-approved, triple GLP-1/GIP/glucagon receptor medicine)
Retatrutide (also known as LY3437943) is not FDA-approved. As of April 2026, it is available only through Eli Lilly’s TRIUMPH Phase 3 clinical trial program, and no approved version exists in any pharmacy. Lilly’s first Phase 3 success came from TRIUMPH-4, which reported topline results on December 11, 2025, showing an average of 71.2 lbs of weight loss over 68 weeks in people with obesity and knee osteoarthritis. That result is from a clinical trial — it does not mean the drug is available for prescribing.
The TRIUMPH program includes four registrational trials: TRIUMPH-1 (type 2 diabetes), TRIUMPH-2 (obesity with other conditions), TRIUMPH-3 (obesity with cardiovascular disease), and TRIUMPH-4 (obesity with knee osteoarthritis). Lilly is expected to file for FDA approval after additional Phase 3 data are collected, but no submission timeline has been confirmed. Until an FDA approval is granted and the drug is commercially available, the only legal source is enrollment in a TRIUMPH trial.
What makes retatrutide different from existing GLP-1 medicines? It works on three hormone signals at once — GLP-1, GIP, and glucagon receptors — while approved drugs like tirzepatide act on two (GLP-1 and GIP) and semaglutide acts on one (GLP-1). This triple hormone mechanism may explain the larger average weight loss numbers in trials, but it also introduces a heart rate signal that is unique to retatrutide and not seen with the other approved medicines.
How long do retatrutide side effects last? (prevalence, onset, and duration by symptom)
For most participants in the Phase 2 and Phase 3 trials, GI side effects were strongest during the first one to two weeks after each dose increase and then eased as the body adjusted. Because retatrutide stays in your system for roughly 6 days (the half-life reported by Jastreboff NEJM 2023), it takes about two weeks at each new dose for blood levels to stabilize — which is why the TRIUMPH-4 schedule holds each dose for four weeks before stepping up.
The Phase 2 data (Jastreboff 2023 NEJM) described GI adverse events as generally mild to moderate in severity and linked to the dose taken. The heart rate increase was different — it built up over time, peaking around week 24 before starting to decline. Participants who progressed to the 12 mg dose in TRIUMPH-4 had an 18.2% rate of discontinuing due to adverse events, compared to 12.2% at 9 mg and 4.0% on placebo. Those numbers reflect the real tolerability tradeoff at the highest doses.
“This app has been so helpful in helping me understand how my medication builds in my system from week to week. I would've been so lost without the graph”
Is compounded retatrutide safe? (No — it is unapproved and illegal)
If you have seen retatrutide advertised for sale online or from a compounding pharmacy, that product is not legal. Retatrutide has never been FDA-approved, which means compounding pharmacies cannot legally produce or sell it. The FDA has issued warning letters to at least three sellers for distributing unapproved retatrutide as a new drug.
On March 31, 2026, the FDA sent a warning letter to Mile High Compounds LLC for selling unapproved new drugs including retatrutide. On September 9, 2025, warning letters went to both GLP-1 Solution and Amazing Meds for the same practice. The FDA’s broader guidance on unapproved GLP-1 drugs makes clear that products sold as peptides outside of an FDA-approved pathway are illegal and have no quality control, safety testing, or verified purity. Health Canada and the UK’s MHRA have issued similar advisories about unauthorized injectable peptide drugs.
Unlike compounded semaglutide or tirzepatide (which were made legal during shortage periods under specific FDA rules), there is no shortage exception for retatrutide because no approved version of retatrutide exists. This is an important distinction: the rules that temporarily allowed compounded versions of Wegovy and Mounjaro during supply shortages simply do not apply to a drug that has never been approved. Any product sold as retatrutide outside a Lilly trial is an unapproved new drug under federal law.
!Never buy compounded retatrutide
Compounded retatrutide is illegal under FDA rules. No approved version exists, so no compounding exception applies. The FDA has issued warning letters to sellers. Quality, purity, and dosing are completely unverified. The side-effect data from TRIUMPH trials does NOT apply to any compounded product.
Which retatrutide side effects are serious? (pancreatitis, gallbladder, thyroid, vision)
Most side effects in retatrutide trials were uncomfortable but not dangerous. A small group are red flags that require same-day medical attention. Because no FDA label exists, the serious-risk profile is inferred from the GLP-1 class of medicines and from what was monitored in Phase 2 and Phase 3 trials.
Pancreatitis is the most important to know about. GLP-1 medicines as a class have been associated with a small increase in pancreatitis risk, and trial protocols for retatrutide monitor for it. Signs: sudden, severe pain in the upper stomach that bores through to your back, especially with nausea or vomiting that will not let up. This is not something to watch and wait on — it needs same-day emergency evaluation.
Gallstones are a risk tied to rapid weight loss rather than the drug specifically. When weight drops quickly — from any cause — the liver releases more cholesterol into bile, raising the chance of stones forming. Trial participants who lost an average of 71.2 lbs over 68 weeks in TRIUMPH-4 are in the rapid-loss category. Sharp pain in the upper right abdomen, especially after eating, is worth a call to your trial team.
Thyroid cell warnings are a class-level concern for GLP-1 medicines based on rodent studies showing thyroid C-cell tumors. No FDA label exists to state a specific warning for retatrutide, but GLP-1 class warnings likely apply. If you notice a new lump in your neck or have a family history of medullary thyroid cancer or MEN 2 (a rare inherited hormone condition), tell your trial physician before starting.
Sudden vision change on any medication is worth attention. The European Medicines Agency’s safety review committee concluded in June 2025 that nonarteritic anterior ischemic optic neuropathy (NAION) — a rare condition that can cause sudden vision loss in one eye — is a very rare side effect of semaglutide medicines specifically (Ozempic, Wegovy, Rybelsus, oral Wegovy). No NAION regulatory signal has been identified for retatrutide by any regulator, and no FDA label exists because retatrutide is investigational. Even so, if you experience a sudden change in vision or partial vision loss on one side during a trial, seek medical evaluation the same day and tell your trial team.
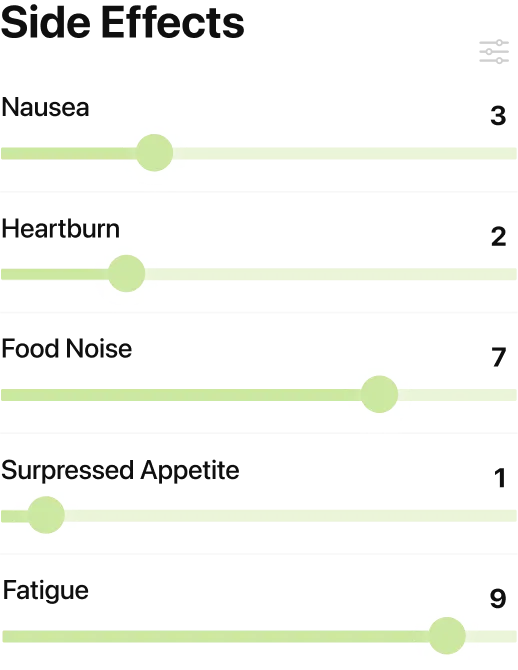
!Call your trial team or go to urgent care right away if you notice
- Severe stomach pain that won’t go away
- pain that spreads to your back
- vomiting lasting more than 24 hours
- yellow skin or eyes
- a rapid or irregular heartbeat that feels new
- a new lump in your neck
- trouble breathing or swallowing.
How does retatrutide affect heart rate?
One of the notable signals in the Phase 2 trial was how heart rate responded to each step-up. Jastreboff et al. (NEJM 2023) reported that resting pulse climbed as doses went up, peaked at around week 24, and then started to decline. This pattern is linked to the glucagon arm of the triple mechanism — glucagon naturally raises cardiac output, so a medicine that activates that pathway alongside GLP-1 tends to push resting pulse up more than GLP-1-only medicines.
This is different from what happens with tirzepatide or semaglutide, both of which show small heart rate increases in trials but without the same glucagon-driven pattern. Trial protocols for retatrutide include cardiovascular monitoring specifically because of this signal. If you are in a TRIUMPH trial and notice that your resting heart rate has risen persistently or that you feel palpitations, report it to your trial team — that is exactly the kind of signal the trial is designed to capture and monitor.
There is no verified specific number to cite here. Secondary sources have reported various bpm figures, but the primary trial publications do not provide a single verified value that is publicly accessible in full-text form. What is confirmed from the published NEJM abstract and results: the heart rate effect grew with each dose step, peaked around week 24, and then declined. Anyone with pre-existing heart rhythm problems should discuss this specifically with their trial physician before participating.
What’s the best way to manage nausea on retatrutide?
The three tactics that work for most people on weekly GLP-1 medicines: eat small protein-first meals on shot day, stay hydrated, and talk to your trial team about slowing your dose pace if nausea is severe. At 43.2% at the 12 mg dose in TRIUMPH-4, nausea is the most common side effect — but it is also the one with the most practical management options.
Because retatrutide stays in your system for roughly 6 days, your body is adjusting to elevated drug levels for most of the week after a shot, not just for 24 to 48 hours. That means the usual strategy of managing nausea only on shot day is not enough — protein intake and hydration matter across the full week. A protein target of roughly 1.2 to 1.6 grams per kilogram of body weight per day (the range commonly recommended for clinical weight loss in published nutrition guidance), spread across smaller meals, keeps your stomach from being empty while also protecting lean mass.
“The app is great, tracks everything. Scan your food and drink and it’s there on your list. I check it multiple times a day to track my steps. I add my water and the app lets me know where I am with protein and fiber. Add a Hume or other 8 pt scale will add towards completing your weight loss goal. Love how the app tracks my glp1 and how much I have each day!”
Ginger and peppermint have published evidence for mild nausea relief and are both low-risk additions to a GLP-1 routine. Ginger tea or candies work well in the 24 to 48 hours after each injection. If queasiness is severe enough to prevent you from eating or drinking, tell your trial team — anti-nausea medication is sometimes provided in trial protocols.
Does retatrutide cause diarrhea?
Yes. Diarrhea affected 33.1% of participants at the 12 mg dose and 34.7% at 9 mg in TRIUMPH-4 (versus 13.4% on placebo), making it the second most common GI side effect. It is almost always mild and usually starts in the first one to two weeks after starting or stepping up. The mechanism is the same as with other GLP-1 medicines: slowed stomach emptying and changes in gut motility.
The most practical management approach: stay hydrated (at least 64 oz of water daily), avoid very high-fat meals and foods with sugar alcohols like sorbitol or xylitol (common in protein bars and sugar-free snacks), and stick to a bland diet — rice, bananas, toast, boiled potatoes — when diarrhea is active. If it is bloody, lasts more than three days, or comes with signs of dehydration like dark urine or dizziness, contact your trial team.
Does retatrutide cause constipation?
Yes. Constipation affected 25.0% of participants at 12 mg and 21.8% at 9 mg in TRIUMPH-4 (versus 8.7% on placebo). The cause is the same as with other GLP-1 medicines: slowed digestion means slower transit time, which dries out stool. The fix is consistent rather than dramatic — more water, more fiber (25 to 35 grams a day from vegetables, legumes, chia seeds, and ground flax), and daily walking.
A practical approach is to build fiber up gradually, adding one high-fiber food per meal every few days rather than jumping to 35 grams overnight, which often causes bloating. If dietary changes are not enough, ask your trial team or a pharmacist about an over-the-counter stool softener. If you go more than three days without a bowel movement or develop severe pain, contact your trial team.
Does retatrutide cause vomiting or abdominal pain?
Vomiting affected 20.9% of participants at the 12 mg dose and 20.4% at 9 mg in TRIUMPH-4, versus 0.0% on placebo. That is a meaningful jump from placebo and sits between the rates reported for tirzepatide 15 mg in SURMOUNT-1 (12.2%) and semaglutide 2.4 mg in STEP 1 (24.8%). Most vomiting episodes in GLP-1 trials are isolated rather than persistent and tend to cluster in the first week after each dose step-up.
The main risk from vomiting is dehydration. If you cannot keep fluids down for more than 24 hours, contact your trial team or seek care that day. Eating small meals every three to four hours — rather than waiting for hunger, which the medicine suppresses — is the most effective way to prevent the empty-stomach nausea that often triggers vomiting on weekly GLP-1 medicines.
Does retatrutide cause headaches or fatigue?
Fatigue and headaches are common across the GLP-1 medicine class and appear in the Phase 2 retatrutide data as part of the general tolerability profile. Both are usually mild and cluster in the 48 to 72 hours after each injection when medicine levels peak. The most common underlying cause is dehydration or low blood sugar from eating much less than usual — the medicine suppresses appetite effectively enough that some people skip meals without realizing it.
The practical fix: set a daily water target and treat it as non-negotiable (the medicine blunts thirst signals, so waiting until you feel thirsty is too late), eat protein-containing food even when you are not hungry, and avoid scheduling hard workouts the day after your shot during the dose step-up months. Fatigue typically improves after weight begins to stabilize and the body adjusts to the new eating pattern.
Does retatrutide cause hair loss?
Hair shedding is a class effect of rapid weight loss rather than a drug-specific reaction, and it is relevant for retatrutide because of the large average weight loss seen in trials. In TRIUMPH-4, participants lost an average of 71.2 lbs over 68 weeks — that rate of weight loss is in the range where stress-triggered shedding (called telogen effluvium in dermatology) typically occurs.
The mechanism: any fast, sustained weight loss pushes a larger share of hair follicles into their resting phase at the same time, and about three months later those hairs fall out together. The reassuring part is that this is almost always reversible. Once weight stabilizes, follicles cycle back to normal. Dermatology literature on stress-triggered shedding puts typical resolution in the six to twelve month window after the triggering stress settles.

Two things reduce the severity based on general dermatology guidance: adequate protein intake (published nutrition guidance for clinical weight loss typically recommends 1.2 to 1.6 grams per kilogram of body weight per day) and avoiding very low calorie days. The body prioritizes vital organs over hair follicles when it is running low on both calories and protein. Tracking protein daily — not just guessing — is the most actionable change most people can make.
Are retatrutide side effects worse for women?
Published sex-subgroup analyses of GLP-1 medicines consistently show women reporting GI side effects — nausea, vomiting, constipation — at slightly higher rates than men, though the gap is smaller than many headlines suggest. The same pattern is expected for retatrutide based on the class-level data, though TRIUMPH-4 topline results did not include a sex-stratified breakdown.
Hair shedding tends to be more noticeable in women than men, both because women tend to have longer hair and because the TRIUMPH-4 trial produced a rate of weight loss large enough to trigger telogen effluvium in many participants. The mechanism is tied to the rate of weight loss, not the sex of the person, but women often notice it more visibly.
Retatrutide is not recommended during pregnancy or breastfeeding. GLP-1 class warnings advise stopping the medicine at least two months before a planned pregnancy because of the drug’s long stay in the body (roughly 6 days to drop by half). If you become pregnant during a TRIUMPH trial, contact your trial team immediately — the protocol will guide what to do next.
What about pregnancy and fertility on retatrutide?
Retatrutide is not recommended during pregnancy. TRIUMPH trial protocols exclude people who are pregnant, and GLP-1 class warnings based on animal studies advise stopping the medicine before conception. Because retatrutide takes about six days to drop by half in the body, clinical trial protocols typically require stopping well in advance of any planned pregnancy.
Because retatrutide has no FDA label and has not been approved, there is no human safety data on pregnancy outcomes for retatrutide specifically. If you are considering a TRIUMPH trial and are of childbearing age, the trial consent process will walk you through the specific requirements and contraception recommendations. This is a question to ask your trial team directly rather than relying on guidance meant for approved GLP-1 medicines.
What about injection site reactions?
Injection site reactions — redness, mild swelling, itching, or a small bruise — were reported in Phase 2 but were not a dominant adverse event. They are typical for any weekly shot medicine and almost always mild. Rotating your injection site across your stomach, outer thigh, and upper arm with each dose reduces localized irritation.
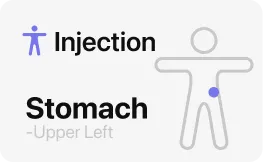
If you notice a hard lump that does not go away after a week, spreading redness that grows over 24 hours, warmth, pus, or fever at the injection site, contact your trial team — signs of infection at an injection site need medical evaluation and are not something to wait out.
Separate from a localized injection-site reaction is a broader skin sensation signal that stood out in TRIUMPH-4: dysesthesia (abnormal skin sensations such as tingling, burning, or prickling) was reported in 20.9% of participants at the 12 mg target dose versus 0.7% on placebo. Phase 2 (Jastreboff 2023 NEJM) also noted cutaneous hyperesthesia and skin sensitivity at roughly 7% on retatrutide versus 1% on placebo. These events were generally mild, did not always cluster around the shot itself, and most resolved during the trial. If you are in a TRIUMPH study and you develop tingling, burning, or numbness that persists or spreads, flag it to your trial team so it can be documented and monitored.
What happens if you stop retatrutide?
If you stop retatrutide, stomach side effects resolve as the medicine clears your system — typically within two to three weeks, given that it takes about 6 days to drop by half. Nausea, vomiting, and fatigue end first. Constipation may linger slightly longer as gut motility returns to normal.
The more challenging part is appetite returning. GLP-1 class data consistently shows that hunger comes back within one to two weeks of stopping, and body weight tends to return over the following months. The STEP 4 trial (semaglutide) and SURMOUNT-4 (tirzepatide) both demonstrated significant weight regain after discontinuation. This class pattern is expected to apply to retatrutide as well. If you are in a TRIUMPH trial and thinking about stopping, talk to your trial team about what the protocol recommends rather than stopping abruptly.
Retatrutide vs tirzepatide vs semaglutide side effects
All three medicines share the same GI side-effect profile because they all act on GLP-1 receptors: nausea, diarrhea, constipation, and vomiting are the most common on each. Retatrutide’s TRIUMPH-4 nausea rate (43.2%) lands close to semaglutide 2.4 mg’s STEP 1 rate (44.2%) and above tirzepatide 15 mg’s SURMOUNT-1 rate (31.0%). The heart rate signal is unique to retatrutide in the current landscape — no comparable dose-driven pulse rise has been reported for the two approved medicines.
| Side effect | Retatrutide 12 mg (TRIUMPH-4) | Tirzepatide 15 mg (SURMOUNT-1) | Semaglutide 2.4 mg (STEP 1) |
|---|---|---|---|
| Nausea | 43.2% | 31.0% | 44.2% |
| Diarrhea | 33.1% | 23.0% | 31.5% |
| Vomiting | 20.9% | 12.2% | 24.8% |
| Constipation | 25.0% | 11.7% | 23.4% |
| Heart rate increase | Grows with dose, peaks ~24 wks | Small, class-level | Small, class-level |
Retatrutide: Phase 3 TRIUMPH-4 topline (Eli Lilly press release, December 11, 2025). Tirzepatide: SURMOUNT-1 (Jastreboff et al., NEJM 2022) — Supplementary Appendix safety tables at the 15 mg dose. Semaglutide: STEP 1 (Wilding et al., NEJM 2021) — Table 2 adverse-event counts, 2.4 mg arm (n=1306). Rates are not directly comparable across trials (different populations, designs, follow-up) — retatrutide’s rates look close to semaglutide’s on nausea and diarrhea despite higher weight loss.
An important practical point: if you are comparing these medicines because you are trying to decide what to discuss with your doctor, tirzepatide (Zepbound/Mounjaro) and semaglutide (Wegovy/Ozempic) are FDA-approved and available today. Retatrutide is not. The only way to access retatrutide now is enrollment in a TRIUMPH clinical trial.
How do the TRIUMPH Phase 3 dose steps work? (2 → 4 → 6 → 9 → 12 mg)
In TRIUMPH-4, participants started at 2 mg once weekly and stepped up every four weeks in a set sequence: 2 mg for four weeks, then 4 mg for four weeks, then 6 mg for four weeks, then 9 mg (target). People assigned to the 12 mg maximum target took one more step after 9 mg. Each level was held for four weeks before moving up, following the same logic as tirzepatide’s schedule — give the body enough time at a stable level before adding more. Condensed: 2 → 4 → 6 → 9 mg to the 9 mg target, or 2 → 4 → 6 → 9 → 12 mg for the maximum 12 mg target.
Each dose step-up briefly resets the GI side-effect curve. Most participants found nausea and diarrhea peaked in the first one to two weeks at the new dose and then eased. The pattern typically got milder with each successive step-up as the body became more familiar with the drug. The practical implication: the 2 to 4 mg step-up may feel harder than expected, but by the time you are stepping from 9 to 12 mg, most people’s tolerability has improved substantially.
The 12 mg dose in TRIUMPH-4 had a higher discontinuation rate due to adverse events (18.2%) than the 9 mg dose (12.2%), which reflects the real tolerability tradeoff at the maximum dose. Whether you reach 9 mg or 12 mg in a trial depends on the protocol and how your body responds — trial teams monitor this closely.
What about alcohol on retatrutide?
No specific alcohol interaction data exists for retatrutide. Based on the class-level mechanism — GLP-1 medicines slow stomach emptying — alcohol is expected to sit longer in the stomach than usual, which means it can hit harder with fewer drinks. Most people on GLP-1 medicines report that their alcohol tolerance drops, and that nausea after drinking becomes more pronounced. The practical guidance used across GLP-1 medicines applies here: eat a protein-containing meal before drinking, stick to lower amounts than you normally would, and avoid alcohol in the 48 hours after each dose step-up when stomach side effects are already at their peak.
Should you wait for retatrutide or start an approved GLP-1 now?
Starting an approved GLP-1 now is usually the right call if you qualify and can access one. Retatrutide’s FDA approval timeline has not been publicly committed by Eli Lilly, and even an optimistic submission path would put commercial availability at least another year out after a successful New Drug Application review. In the meantime, two FDA-approved drugs with years of real-world data are available today: tirzepatide (Zepbound for obesity, Mounjaro for type 2 diabetes) and semaglutide (Wegovy for obesity, Ozempic for type 2 diabetes). Both have trial-proven weight loss effects, understood safety profiles, and can be prescribed by any licensed clinician. If retatrutide does get approved, switching from an approved GLP-1 is a conversation for a later date.
The one reason to specifically wait is if you have already tried tirzepatide or semaglutide and did not respond well. Retatrutide’s three-target mechanism is different enough that a non-responder on tirzepatide or semaglutide might respond differently — but that is a conversation with your doctor, not a reason to buy compounded retatrutide online. A TRIUMPH trial enrollment is the only legitimate way to access the medicine today.
What about long-term use of retatrutide?
Long-term safety data for retatrutide is limited because the drug is still in Phase 3 trials. The longest data available is 68 weeks from TRIUMPH-4. The question of what happens beyond 68 weeks — including long-term cardiovascular outcomes, sustained weight maintenance, and extended tolerability — will be answered by ongoing trial phases and, eventually, post-marketing surveillance if the drug receives FDA approval.
The heart rate signal is the area that long-term follow-up data will be most important to clarify. Jastreboff 2023 NEJM showed the rise peaked at about week 24 and declined thereafter, but whether it returns fully to baseline and what the implications are for people with pre-existing heart conditions are questions that Phase 3 and eventual real-world data will need to address.
One MeAgain reviewer, Stefanig31 (April 13, 2026), summed up the tracker value of the app independent of which GLP-1 you end up on: “It tracks all your food intake and macros, your activity level, medication level, side effects. 100% recommended.” That same weekly routine — shot day, side-effect rating, food, water, weight — is what a TRIUMPH trial participant needs to keep consistent, and what anyone watching the retatrutide data can practice today on a medicine they already take.
“If you are in your GLP-1 journey, this app will be your best friend. It tracks all your food intake and macros, your activity level, medication level, side effects. 100% recommended.”